Wolff–Kishner Reduction
Definition: What is Wolff-Kishner Reduction?
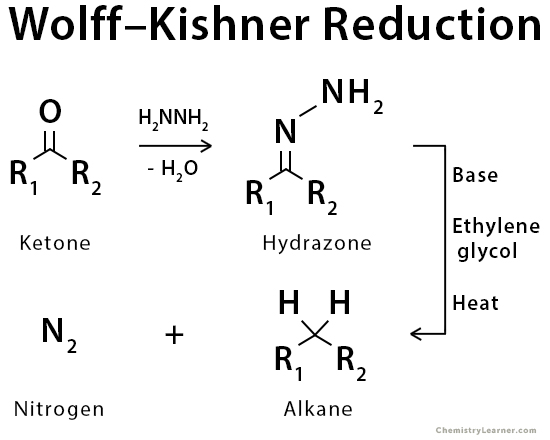
The Wolff-Kishner reduction is an organic reaction used to convert an aldehyde or ketone to an alkane using hydrazine, base, and heat (~200 °C). Aldehydes and ketones are first converted to a hydrazine derivative by reaction with hydrazine. These hydrazones can be further converted to the corresponding alkane by reaction with base and heat. The reaction converts carbonyl groups to methylene groups.
The Wolff-Kishner reduction represents a general method for converting aldehydes and ketones into alkanes. Typically, a high boiling point solvent, such as ethylene glycol, is used to provide the high temperatures needed for this reaction to occur. The driving force for the reaction is the conversion of hydrazine to nitrogen gas. This reaction is used for the reduction of aldehydes and ketones. Other reducible groups, such as carbon-carbon double bonds and nitro groups, usually remain unaffected under the conditions of this reaction [1-5].
The history of Wolff-Kishner reduction goes back more than 100 years when Russian chemist Nikolai Kishner and German chemist Ludwig Wolff independently discovered the reaction in 1911 and 1912, respectively [4].
Examples of Wolff-Kishner Reduction
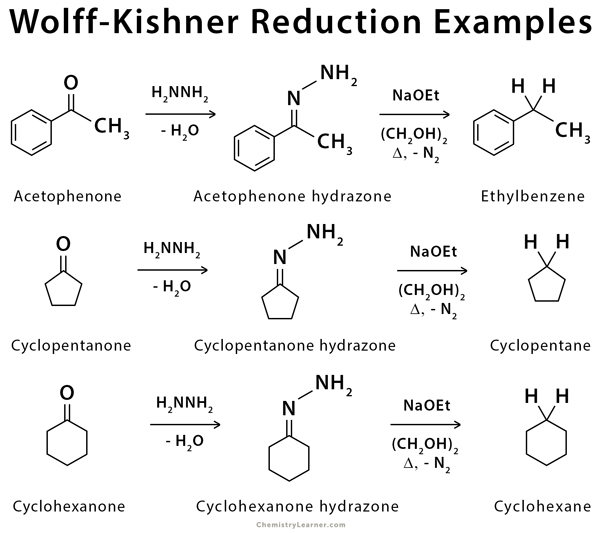
An example of Wolff-Kishner reduction, applicable to aromatic compounds, includes the conversion of cyclohexanone to cyclohexane [1-4,6,7].
Mechanism of Wolff-Kishner Reduction
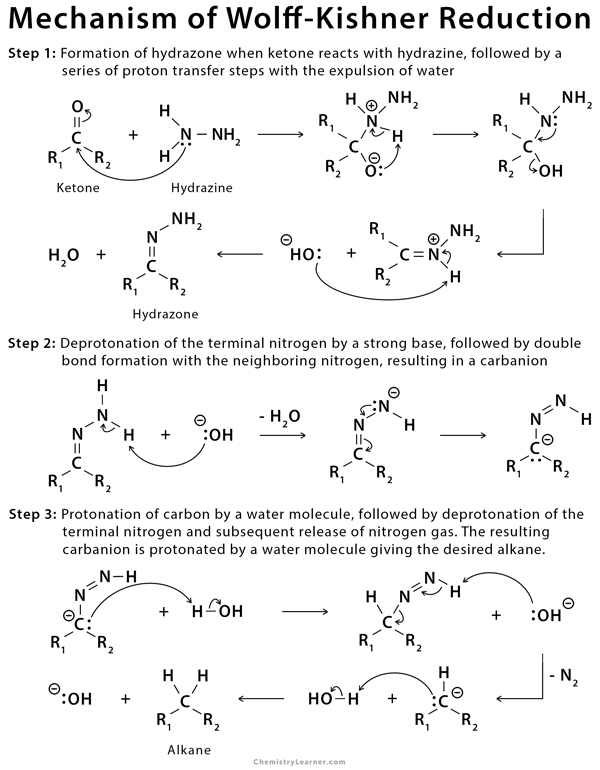
Condensation of the carbonyl compound with hydrazine forms the hydrazone, and treatment with base induces the reduction of the carbon coupled with oxidation of the hydrazine to gaseous nitrogen, to yield the corresponding alkane [1-6,8,9].
Applications of Wolff-Kishner Reduction
Wolff-Kishner reduction has a broad range of applications [10]. Some of them include,
- Organic synthesis of multiwalled carbon nanotube
- Synthesis of functionalized imidazole substrate
Limitations of Wolff–Kishner Reduction
There are three significant limitations of the Wolff-Kishner reduction.
- The reduction requires a high temperature.
- It cannot be used for sterically hindered ketones.
- Although the process is specific for carbonyl groups, sometimes migration of the double bond takes place instead of the expected mechanism of the reaction when an undesired product like alpha, beta-unsaturated carbonyl compounds are formed in the reaction.
Clemmensen Reduction vs. Wolff-Kishner Reduction
Wolff-Kishner reduction is complementary to Clemmensen reduction, which also converts aldehydes and ketones to hydrocarbons.
When to use Wolff-Kishner reduction and Clemmensen reduction?
Wolff-Kishner reduction is better to use for acid-sensitive compounds. As it is a reduction under basic condition, the compound is not affected. The Clemmensen reduction is performed in strongly acidic conditions. If a protecting group is present that can be removed with an acid, such as an acetal or silyl ether, then consider this reduction as an alternative.
Clemmensen Reduction vs. Wolff-Kishner Reduction
Clemmensen Reduction | Wolff-Kishner Reduction |
Carried out in a strongly acidic medium | Carried out in a strongly basic medium |
Hydroxide (OH) is attacked | Hydroxide (OH) is saved and regenerated |
References
- Definition, example, and mechanism – Chem.libretexts.org
- Definition, example, and mechanism – Ochempal.org
- Definition, example, and mechanism – En.chem-station.com
- Definition, example, mechanism, and history – Thieme.de
- Definition and mechanism – Name-reaction.com
- Example and mechanism – Organic-chemistry.org
- Definition and example – Chem.ucla.edu
- Mechanism – Chemtube3d.com
- Mechanism – Ursula.chem.yale.edu
- Applications – Onlinelibrary.wiley.com