Vapor Pressure
Vapor pressure is the pressure exerted by a gas when it is in equilibrium with its liquid or solid form. In a closed container, some molecules of a liquid always escape into the air as gas, while some gas molecules return to the liquid. When the number of molecules leaving the liquid equals the number returning, the system is in equilibrium, and the vapor pressure remains constant. [1-4]
Vapor Pressure Formula
Vapor pressure can be calculated using different formulas depending on the situation. It depends on temperature and the nature of the liquid. Two important equations help describe vapor pressure: Raoult’s Law and the Clausius-Clapeyron equation. [1-5]
Raoult’s Law
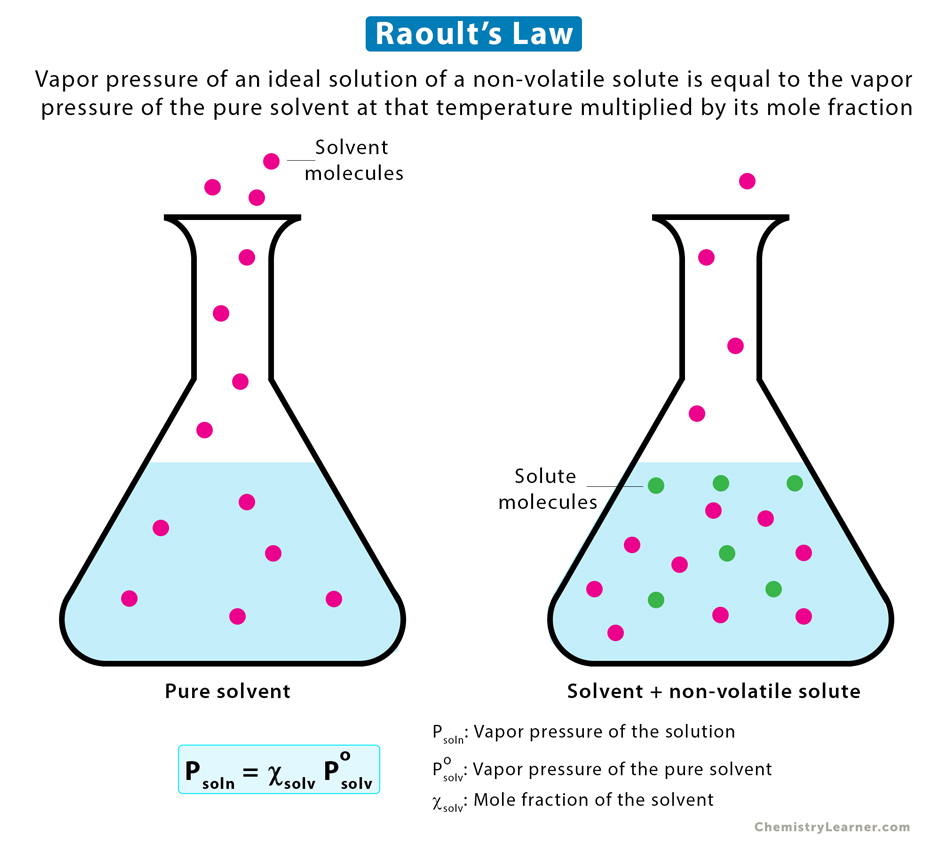
Raoult’s Law describes how adding a solute (such as salt or sugar) to a liquid lowers its vapor pressure. The law states that the vapor pressure of a solution (Psoln) is lower than that of the pure solvent (Psolv) and depends on the mole fraction (Χsolv) of the solvent:
Psoln = Χsolv Psolv
It means that when a non-volatile solute (one that doesn’t evaporate) is added to a liquid, fewer molecules can escape into the gas phase, reducing the vapor pressure. This is why saltwater evaporates more slowly than pure water.
While Raoult’s Law explains the effect of solutes, the Clausius-Clapeyron equation describes how temperature affects vapor pressure.
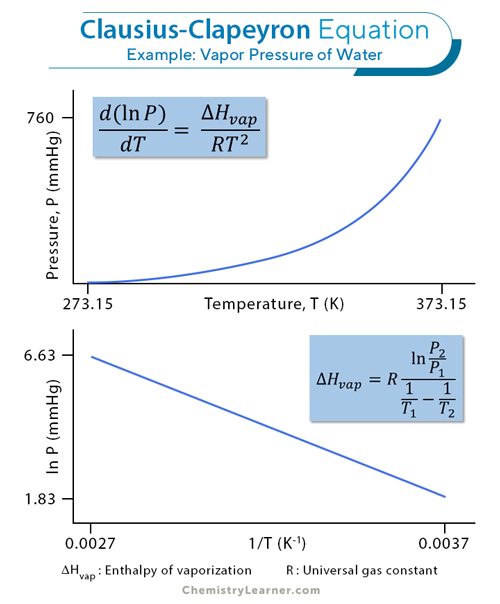
The Clausius-Clapeyron equation shows how the vapor pressure of a substance increases with temperature. It is written as:
ln (P2/P1) = (ΔHvap /R)(1/T1 – 1/T2)
Where:
– P1: Vapor pressure at temperature T1
– P2: Vapor pressure at temperature T2
– ΔHvap: Molar enthalpy (heat) of vaporization
– R: Universal gas constant
– T: Absolute temperature in Kelvin
This equation explains why liquids evaporate faster at higher temperatures. As temperature increases, molecules gain more energy to escape into the gas phase, increasing vapor pressure. For example, water evaporates faster on a hot day than on a cold day because its vapor pressure is higher.
Factors Affecting Vapor Pressure
Two major factors influence vapor pressure: the nature of the liquid and temperature. [1-5]
1. Nature of the Liquid
Different liquids have different vapor pressures depending on the strength of their intermolecular forces. If strong forces hold together the molecules in a liquid, they find it harder to escape into the air, leading to a lower vapor pressure. On the other hand, if the forces between molecules are weak, they escape more easily, resulting in a higher vapor pressure.
For example, water has strong hydrogen bonds between its molecules, so it has a lower vapor pressure and evaporates more slowly. In contrast, acetone (found in nail polish remover) has weaker forces between its molecules, allowing it to evaporate quickly and have a higher vapor pressure. This is why acetone dries much faster than water when left in an open container.
2. Temperature
Temperature has a direct effect on vapor pressure. As temperature increases, the molecules in a liquid gain more kinetic energy and move faster. This allows more molecules to escape into the gas phase, increasing the vapor pressure.
A simple example is boiling water. At room temperature, water has a relatively low vapor pressure, but when it is heated, the vapor pressure rises. Water boils when its vapor pressure equals the surrounding atmospheric pressure. This is why water boils at 100°C at sea level, but at a lower temperature in high-altitude places where atmospheric pressure is lower.
A graph of vapor pressure vs. temperature shows a steep curve that rises as temperature increases. This means that even a small increase in temperature can cause a large increase in vapor pressure, which is why liquids evaporate faster on hot days.
Units of Vapor Pressure
Vapor pressure is measured in different units depending on the context. The most commonly used units are: [1-5]
- Pascal (Pa): The SI unit of pressure, where 1 Pa = 1 N/m².
- Torr (or mmHg): Commonly used in chemistry, where 1 Torr = 1 mmHg (millimeter of mercury). Standard atmospheric pressure is 760 Torr.
- Atmosphere (atm): Used in general pressure measurements, where 1 atm = 101.325 kPa = 760 mmHg.
- Bar: Another unit used in physics and engineering, where 1 bar = 100 kPa.
For example, the vapor pressure of water at 25 °C is 23.8 mmHg or 0.0313 atm or 23.8 torr or 3.17 kPa.
Vapor Pressure Table [2]
| Substance | Vapor Pressure at 25 °C (atm) |
|---|---|
| Diethyl ether | 0.7 |
| Bromine | 0.3 |
| Ethyl alcohol | 0.08 |
| Water | 0.03 |
Examples of Vapor Pressure [1-5]
- Boiling Water: At sea level, water boils at 100°C because the atmospheric pressure is high. In high-altitude areas, such as mountains, atmospheric pressure is lower, so water boils at a lower temperature.
- Rubbing Alcohol: When you apply rubbing alcohol on your skin, it evaporates quickly and makes your skin feel cool. This happens because alcohol has a higher vapor pressure than water, meaning it turns into a gas more easily.
- Perfume Diffusion: When you spray perfume, it quickly evaporates because it contains volatile liquids with high vapor pressure. This allows the scent to spread through the air.
- Gasoline Evaporation: Gasoline has a high vapor pressure, which easily allows it to evaporate when exposed to air. This is why you often smell gasoline fumes near a fuel station or when refueling a car.