Vanadium
What is Vanadium
Vanadium (pronunciation: veh-NAY-dee-em) is a medium-hard, silvery element belonging to the family of transition metals represented by the chemical symbol V [1, 2]. Because of its malleability, ductility, and corrosion-resistant properties, vanadium is increasingly used in the manufacturing industry [2].
Its Isotopes
It has two naturally occurring isotopes, out of which 51V is stable while 50V is radioactive with a half-life period of about 1.4 X 1017 years [1]. There are 24 artificial radioisotopes with mass numbers ranging from 40 to 65, 49V and 48V being the two most stable of them with half-life periods of 330 days and 15.9735 days respectively [3].
Where is Vanadium Found
Although it is rarely found free in nature, vanadium can be extracted from 65 different mineral ores, including patronite, carnotite, and vanadinite [1]. It is also obtained as organic complexes from some crude oils, iron ores, and phosphate rocks [1, 2]. Vanadium is generally extracted by heating crushed mineral ores along with chlorine and carbon, producing vanadium (III) chloride, which is then reduced with magnesium in a pressure vessel [2].
The top 3 vanadium reserve holding countries are China, Russia, and South Africa while the top 3 producers include South Africa, China, and Russia [1].
History
Origin of its Name: It is named after ‘Vanadis’, the other name for the Norse goddess Freyja associated with beauty, love, fertility, gold, war, and death [1].
Who discovered it: The Spanish-Mexican chemist Andrés Manuel del Rio is known for the discovery of vanadium [1].
When and How was it Discovered
In 1801, the element was first identified in a sample of Pb5(VO4)3Cl or vanadite, by Professor Manuel Del Rio in Mexico City [1]. He sent the specimen to Paris where the French chemist Victor Collet-Descotils examined it and announced that it was actually chromium [1, 4].
In 1831, vanadium was found by the Swedish chemist Gabriel Selfström at the Swedish capital Stockholm [1]. He separated the element from a specimen of cast iron produced from a mineral ore mined at Småland [1]. He also examined another vanadium mineral, obtained from the Mexican town of Zimapan [1].
In 1869, Sir Henry Roscoe produced pure vanadium at Manchester, England and revealed that the previous samples were actually vanadium nitride (VN) [1].
Vanadium Identification |
|||
| Atomic number | 23 [1] | ||
| CAS number | 7440-62-2 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 5 [1] | 4 [1] | d [1] | |
Properties and Characteristics of Vanadium
General Properties |
||||||||||||||||
| Relative atomic mass | 50.942 [1] | |||||||||||||||
| Atomic mass | 50.942 amu [1] | |||||||||||||||
| Molar mass | About 50.9415 g/mol [5] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Silvery [1, 4] | |||||||||||||||
| Melting point/freezing point | 1910 °C, 3470 °F [1] | |||||||||||||||
| Boiling point | 3407 °C, 6165 °F [1] | |||||||||||||||
| Density | 6.0 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 4] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 628 MPa [6] | |||||||||||||||
| – Mohs | 7 [6] | |||||||||||||||
| – Vickers | 628 MPa [6] | |||||||||||||||
| Electrical conductivity | 5X106 S/m [6] | |||||||||||||||
| Charge | +5, +3 [9] | |||||||||||||||
| Thermal (heat) conductivity | 31 W/(m K) [6] | |||||||||||||||
| Specific heat | 489 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | 158 GPa [1] | |||||||||||||||
| Shear modulus | 46.7 GPa [1] | |||||||||||||||
| Young’s modulus | 127.6 GPa [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | – | – | 2.79X 10-10 | 4.35X 10-7 | 1.07X 10-4 | 7.69X 10-3 | 0.233 | 3.68 | 32.6 | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | -1, +1, +2, +3, +4, +5[1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 50V | 49.947 | 0.25 | 1.4 X 1017 y | EC | ||||||||||||
| 51V | 50.944 | 99.75 | – | – | ||||||||||||
Atomic Data of Vanadium (Element 23)
| Valence electrons | 5 [7] | |||||||
| Quantum numbers | ||||||||
| – n | 3 [8] | |||||||
| – ℓ | 2 [8] | |||||||
| – mℓ | 0 [8] | |||||||
| – ms | +1/2 [8] | |||||||
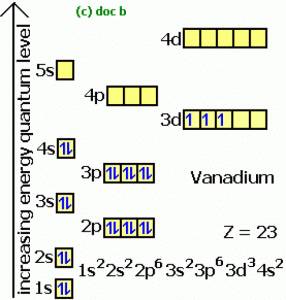
| Electron configuration (noble gas configuration) | [Ar] 3d34s2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 23 [4] | |||||||
| – Number of neutrons | 28 [4] | |||||||
| – Number of protons | 23 [4] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.07 Å [1] | |||||||
| – Covalent radius | 1.44 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 1.63 [1] | |||||||
| Electron affinity | 50.655 kJ mol-1 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 650.908 | 1410.423 | 2828.082 | 4506.734 | 6298.727 | 12362.67 | 14530.7 | 16730.6 | |
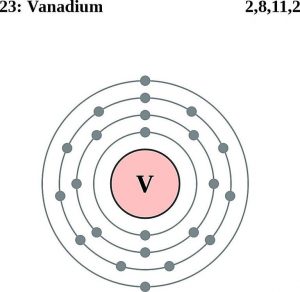
Vanadium Electron Configuration (Bohr Model)
What is It Used for
- About 80 percent of the vanadium manufactured is used for making ferrovanadium, a steel additive [1, 10]. Since vanadium-steel alloys are immensely hard and strong, they are used for producing axles, tools, armor plate, crankshafts, and piston rods [1].
- Adding small amounts of vanadium and chromium (chrome-vanadium), makes steel heat, vibration, and shock resistant [1, 4].
- Because of vanadium’s low parasitic neutron-absorption cross-section, its alloys are used for making the inner structure of nuclear reactors [1].
- An alloy containing vanadium, titanium, and aluminum is used in high-speed aircraft and jet engines [4].
- Vanadium-gallium (V3Ga) superconducting alloys are used for the insert coils of electromagnets while the V3Ga tapes are used in magnets with a field strength of 17.5 teslas [1, 4].
- Vanadium (V) oxide (also called vanadium pentoxide) is used as ceramics and glass coloring pigment, as well as a catalyst for making sulfuric acid [1, 4].
Biological Role in Humans
Aside from some other species, humans need vanadium in trace amounts (0.01 mg) for healthy bone growth and other biological needs [1, 2]. It is obtained from food sources including parsley, mushroom, black pepper, shellfish, dill weed, grains, beer, and wine [2].
Is It Toxic
Consuming or inhaling large amounts of vanadium can be toxic to humans [2]. Lung problems, including pneumonia or bronchitis, may occur in industrial workers exposed to large amounts of vanadium and its compounds [2]. In a study, it has been shown that inhalation of V2O5 causes DNA damage, increasing the risks of cancer in workers.
Interesting Facts
- Body armors and portable artillery pieces used during World War I were made with vanadium [2].
- In a research study, vanadium supplements given to 8 people who have type-2 diabetes for a month helped reduce blood sugar without any serious side effects [2].
- It can withstand attacks by alkalis, salt water, sulfuric and hydrochloric acid [4].
- The element readily oxidizes in air at about 660 °C to form vanadium pentoxide [4].
- The oxidation states of vanadium have green, blue, yellow, and purple colors [2].
- The graphic representation of vanadium indicates the 8th-century figurine of Freyja, the Scandinavian goddess from whom the element’s name is derived [1].
Vanadium Price
The cost of pure vanadium is around $2.20 per gram, and in bulk, its price is around $0.027 [4].
- References
- http://www.rsc.org/periodic-table/element/23/vanadium
- https://www.livescience.com/29155-vanadium.html
- https://education.jlab.org/itselemental/iso023.html
- https://www.chemicool.com/elements/vanadium.html
- https://www.webqc.org/molecular-weight-of-V%28vanadium%29.html
- http://periodictable.com/Elements/023/data.html
- https://www.wou.edu/las/physci/ch462/Vanadium.htm
- http://chemistry-reference.com/q_elements.asp?Symbol=V
- http://www.sciencegeek.net/Chemistry/chempdfs/CommonIons.pdf
- http://www.mining.com/web/vanadium-metal-cant-without-dont-produce/