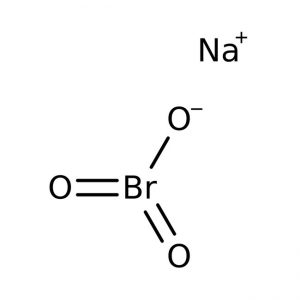
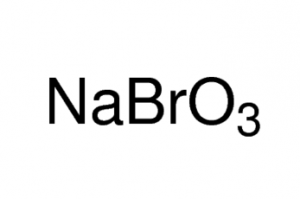Sodium Bromate
Sodium bromate, also called sodium salt of bromic acid, is an inorganic compound of sodium, bromine, and oxygen represented by the chemical formula NaBrO3 [1]. It is a strong oxidizing agent that may explode through prolonged exposure to fire or heat [1].
Sodium Bromate Composition and Synthesis
When bromine reacts with a concentrated, heated solution of sodium carbonate, it produces sodium bromate, sodium bromide, and carbon dioxide as the products [3].
3Br2 + 3Na2CO3 → 5NaBr + NaBrO3 + 3CO2
Sodium bromate is formed through electrolytic oxidation of sodium bromide in which stainless steel plate works as the cathode and mixed metal oxide coated titanium plate acts as the anode [1].
NaBr + 3H2O → NaBrO3 + 3H2
Properties and Characteristics of Sodium Bromate
General Properties |
|
| Molar Mass/Molecular Weight | 150.891 g/mol [1] |
Physical Properties |
|
| Color and Appearance | Colorless to white crystals, dry powder [1] |
| Odor | Odorless [1] |
| Melting Point | 381 °C, 717.8 °F (decomposes with evolution of oxygen) [1] |
| Boiling Point | 1390 °C, 2530 °F [1] |
| Density | 3.34 g cm-3 [1] |
| pH | 5.0-9.0 at 25 °C [4] |
| State of matter at room temperature | Solid [1] |
| Solubility | Soluble in ammonia and insoluble in ethanol [1] |
| Solubility in Water | 27.5 g/100 g at 0 °C
36.4 g/100 g at 20 °C 48.8 g/100 g at 40 °C 62.6 g/100 g at 60 °C 75.8 g/100 g at 80 °C 90.8 g/100 g at 100 °C [1] |
| Magnetic Susceptibility (χ) | -44.2 X 10-6 cm3/mol |
Atomic Properties |
|
| Crystal Structure | Cubic |
Uses
- As a permagent in hair treatment formulations [1].
- Boiler cleaning and dyeing applications that involve oxidation of sulfur dyes [1].
- Combined with sodium bromide as a gold solvent in mines [1].
Is It Safe
Sodium bromate is known to cause acute toxicity by ingestion, inhalation, and through contact with skin and eyes [1]. It causes serious skin irritation, eye damage, and may result in respiratory tract irritation [1]. Being a carcinogen, it may cause chromosomal abnormalities and genetic defects [1].
- References
- Sodium Bromate – Pubchem.ncbi.nlm.nih.gov
- Sodium Bromate – Chemspider.com
- Bromine React With Sodium Carbonate – Chemiday.com
- Sodium Bromate – Sigmaaldrich.com