Home / Physical Chemistry / Sulfur Trioxide (SO3) Formal Charge
Sulfur Trioxide (SO3) Formal Charge
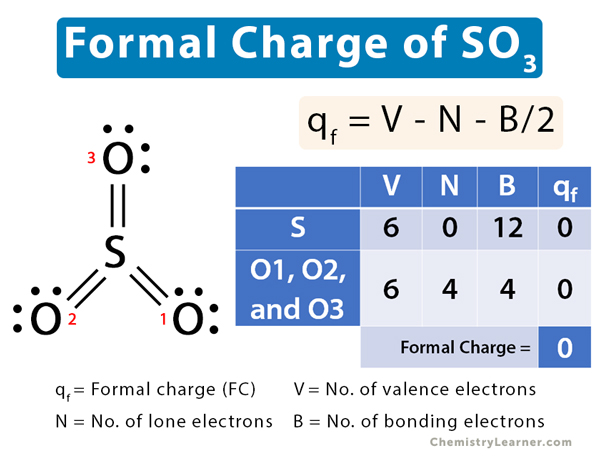
In sulfur trioxide (SO3), sulfur (S) shares double covalent bonds with all oxygen (O) atoms. Its Lewis structure is shown below.
Let us calculate the formal charge of SO3 by calculating the formal charges on S and O.
V = 6, N = 0, B = 12
Therefore,
qf = 6 – 0 – 12/2 = 0
The formal charge on S in SO3 is zero.
Oxygen 1, Oxygen 2, and Oxygen 3
V = 6, N = 4, B = 4
Therefore,
qf = 6 – 4 – 4/2 = 0
The net formal charge is: 0 + 3 x 0 = 0
The formal charge of SO3 is zero. The structure shown above is the most stable.



Nice and clear explanation with few sentences