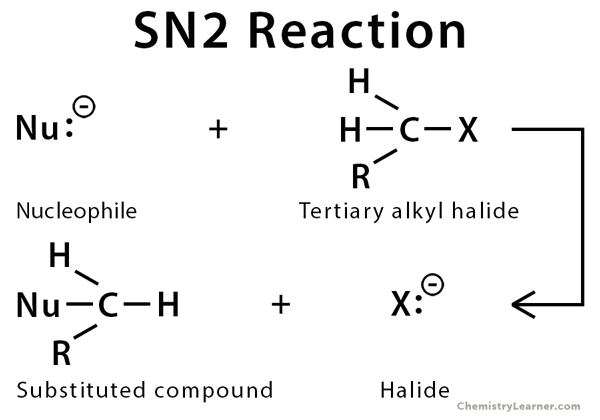SN2 Reaction
Definition: What is SN2 Reaction?
Nucleophiles, typically, have a lone pair of electrons in them. As the name suggests, they are attracted to the nucleus of an atom. The SN2 reaction is a type of nucleophilic substitution in which an electron-rich nucleophile displaces the halogen atom bonded to the central carbon of an alkyl halide molecule. The halide ion that is displaced from the carbon atom is called the leaving group. SN2 reaction takes place at the site of a saturated carbon atom. Some typical nucleophiles are halide (X–), hydroxy group (OH–), alkoxy group (RO–), cyanide ion (CN–), hydrogen sulfide (H2S), ammonia (NH3), and water (H2O) [1-5].
Characteristics of SN2 Reaction
- One-step reaction process, the reaction is concerted: carbon-halogen bond breaking and carbon-nucleophile bond formation takes place at the same time
- Bimolecular and follows second-order kinetics
- Rate of the reaction depends on the concentrations of the substrate (alkyl halide) and the nucleophile
- Has an inversion stereochemistry at the reaction center
- Polar aprotic solvent is used to enhance the reactivity
- Mostly occurs in primary and secondary alkyl halides
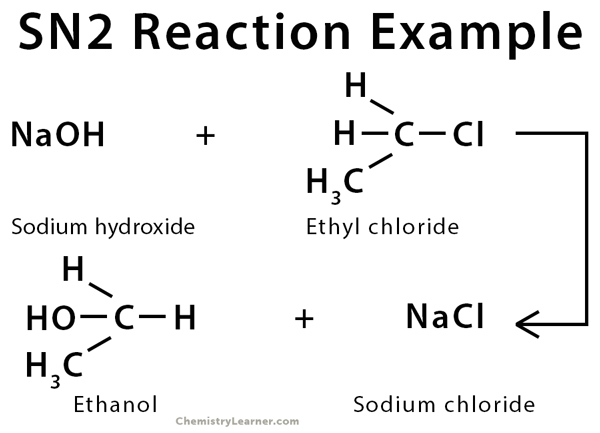
Example of SN2 Reaction
The SN2 reaction is commonly observed in primary and secondary alkyl halides [1].
Factors Affecting SN2 Reaction
SN2 reaction requires to have a good leaving group. Generally, a weak base constitutes an active leaving group because it tends to hold onto its electrons. The order of reactivity of halides towards SN2 reaction is I–>Br–>Cl–>F–. The order of reactivity of alkyl halides in SN2 reactions is methyl > 1° > 2°. 3° alkyl halide does not participate in SN2 reaction because of a steric hindrance (crowded) at the site of reaction. The SN2 reaction is faster in aprotic solvents since the nucleophile attacks the electrophilic carbon and not hydrogen [6-8].
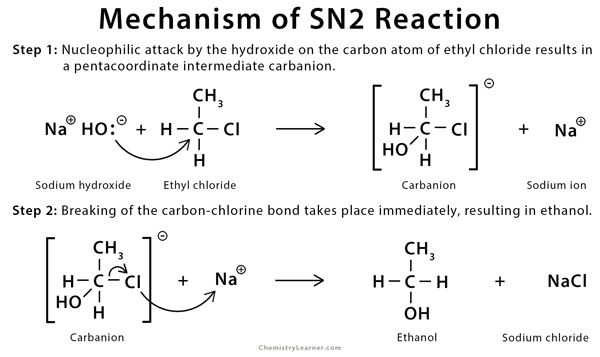
Mechanism of SN2 Reaction
The mechanism of SN2 reaction involves the simultaneous formation of the carbon-nucleophile bond and the breaking of the carbon-halogen bond [1,9,10].
FAQs
Ans. Tertiary alkyl halides cannot use the SN2 mechanism because the presence of alkyl groups surrounding the central carbon atom hinders the nucleophilic attack. Hence, carbon-nucleophile bonce cannot be formed easily.
Ans. SN2 reactions are much slower in polar protic solvents because solvation of nucleophiles by polar protic solvents inhibits the nucleophile’s ability to take part in an SN2 reaction.
Ans. The nucleophilic attack of the SN2 reaction takes place from the backside because the leaving group, which itself is electron-rich, blocks the attack from the front side.
References
- Definition, example and mechanism – Byjus.com
- Definition and example – Masterorganicchemistry.com
- Definition – Chem.libretexts.org
- Definition and mechanism – Chem.ucalgary.ca
- Definition – Chem.ox.ac.uk
- Factors – Iverson.cm.utexas.edu
- Factors – Chem.libretexts.org
- Factors – Chem.libretexts.org
- Mechanism – Chemhelper.com
- Mechanism – Organic-chemistry.org