Schmidt Reaction
Definition: What is Schmidt Reaction?
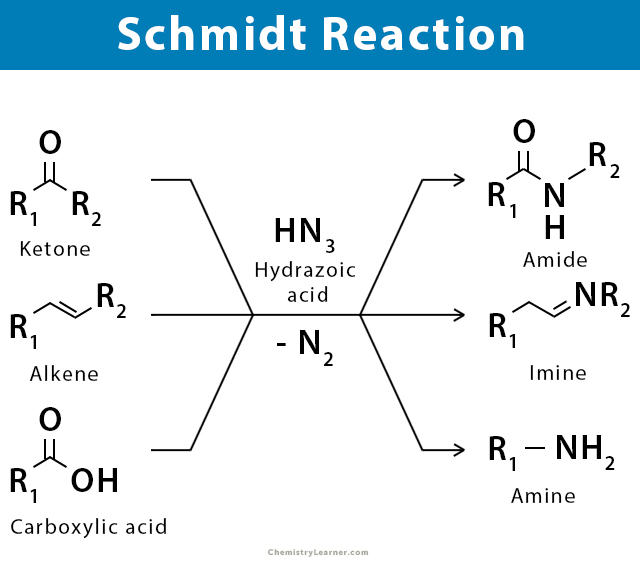
The Schmidt reaction is the reaction of hydrazoic acid or an alkyl azide with a carbonyl compound, alkene, or alcohol, often in the presence of a Brønsted or Lewis acid. The product is an amine, imine, or amide, with the expulsion of nitrogen [1-5].
The history of this reaction goes back to 1924 when German chemist Karl Friedrich Schmidt reported it.
Examples of Schmidt Reaction
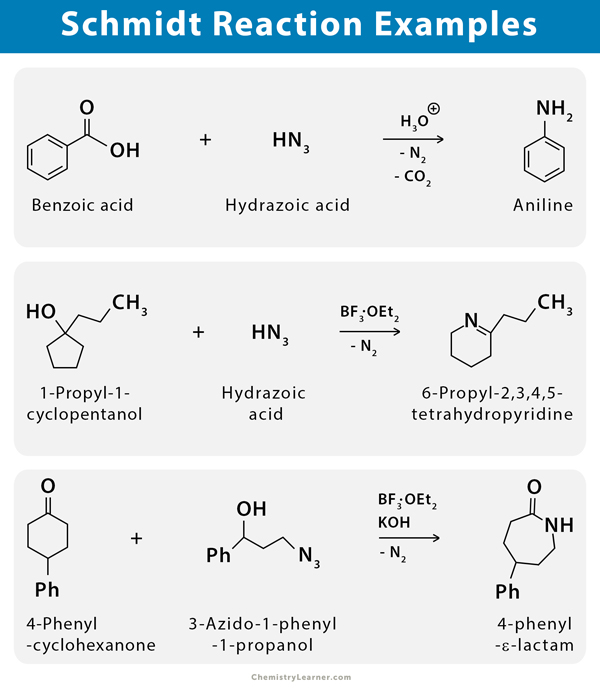
The Schmidt reaction can be used to prepare aniline from benzoic acid and hydrazoic acid [1-3].
Mechanism of Schmidt Reaction
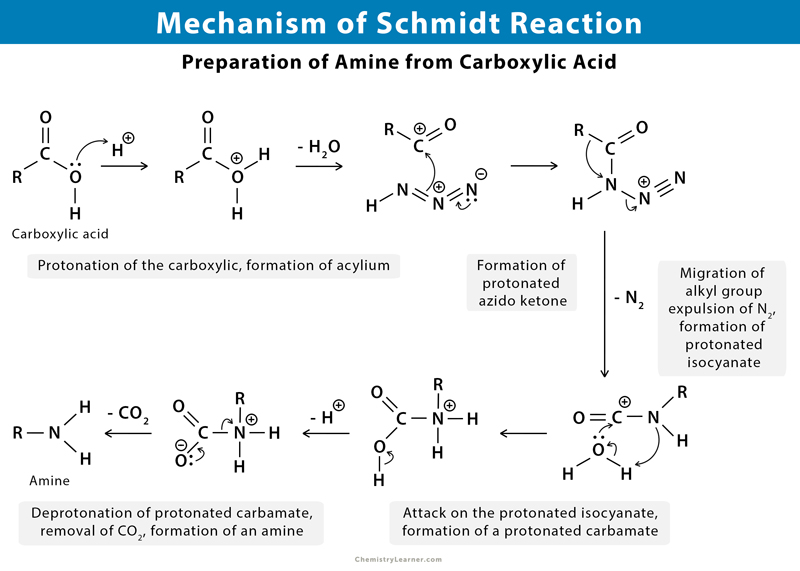
The Schmidt reaction is used to get an amide and an amine by reacting hydrazoic acid to ketone and carboxylic acid, respectively [1-5].
Applications of Schmidt Reaction
The Schmidt reaction can be applied to prepare amino acids, diamines, cyclic amides, lactams, and tetrazole. Each one of these compounds has a unique application. For example, amino acids are used by the industry in the synthesis of drugs and cosmetics [6].
References
- Definition, examples, and mechanism – Onlinelibrary.wiley.com
- Definition, examples, and mechanism – Nature.com
- Definition, examples, and mechanism – Chem.libretexts.org
- Definition and mechanism – Organic-chemistry.org
- Definition and mechanism – Name-reaction.com
- Application – Slideshare.net