Home / Organic Chemistry / Sandmeyer Reaction
Sandmeyer Reaction
Table Of Contents
Definition: What is Sandmeyer Reaction?
Sandmeyer reaction is a type of radical-nucleophilic aromatic substitution reaction. It is a useful tool by which an amino group on an aromatic ring is replaced with different substituents. During the Sandmeyer reaction, the amino group is converted into a diazonium salt that can be transformed into various functional groups using a catalyst. The mechanism involves several steps, including the generation of intermediary compounds [1-3].
Example of Sandmeyer Reaction [4,5]
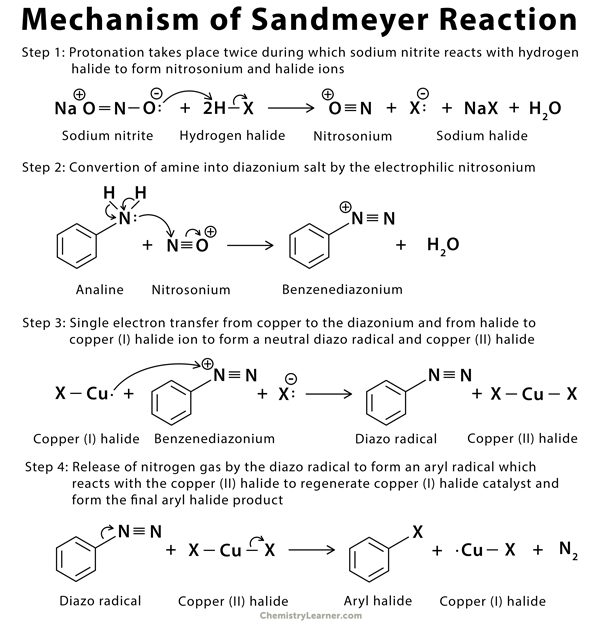
Mechanism of Sandmeyer Reaction [2,3,6,7]
Applications of Sandmeyer Reaction [2]
- Halogenation – Synthesis of aryl halides
- Cyanation – Synthesis of benzonitriles and other useful medicinal drugs
- Trifluoromethylation – Synthesis of aryl compounds having trifluoromethyl group and are used as pharmaceuticals
- Hydroxylation – Conversion of aryl amines into phenols
- References
- Definition – Chem.libretexts.org
- Definition and mechanism – Byjus.com
- Definition and mechanism – Masterorganicchemistry.com
- Examples – Chem.ucla.edu
- Examples – En.chem-station.com
- Mechanism – Name-reaction.com
- Mechanism – Organic-chemistry.org