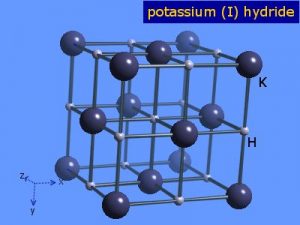
Potassium Hydride
Potassium hydride is an inorganic alkali metal hydride of potassium and hydrogen represented by the chemical formula KH [1]. Since it is an extremely reactive compound, it is commercially available as a suspension of small particles in paraffin wax or mineral oil for easier dispensing [2].
Potassium Hydride Identification |
|
| CAS Number | 7393-26-7 [1] |
| PubChem CID | 82127 [1] |
| ChemSpider ID | 74121 [3] |
| EC Number | 231-704-8 [1, 4] |
| RTECS Number | N/A [4] |
| InChI Key | OCFVSFVLVRNXFJ-UHFFFAOYSA-N [1] |
How is Potassium Hydride Prepared
The synthesis of potassium hydride involves a direct combination of potassium and hydrogen represented by the following equation:
2K + H2 → 2KH
The method of preparation was invented by Sir Humphry Davy after he discovered potassium in 1807 through electrolysis of caustic potash (now called potassium hydroxide) [5].
Reaction with Water
Potassium hydride undergoes a violent reaction with water, producing potassium hydroxide and hydrogen that can burn [6]. It is represented by the following reaction:
KH + H2O → KOH + H2
Properties and Characteristics of Potassium Hydride
General Properties |
|
| Molar Mass/Molecular Weight | 40.106 g/mol [1, 4] |
Physical Properties |
|
| Color and Appearance | White or colorless crystalline solid, gray (commercial sample) [1, 4] |
| Melting Point | ~ 400 °C, 752 °F (decomposes) [3, 7] |
| Boiling Point | N/A [4] |
| Density | 1.47 g cm-3 [4] |
| Charge | 0 (K+ and H–) [1, 3] |
| State of matter at room temperature | Solid [1, 4] |
| Solubility | Insoluble in diethyl ether, benzene, and CS2 [8] |
| Solubility in Water | Decomposes in hot and cold water [8] |
| Heat Capacity (C) | 37.91 J/(mol.K) |
| Acid Dissociation Constant at Logarithmic Scale (pKa) | 35 [9] |
Atomic Properties |
|
| Crystal Structure | Cubic |
Uses
- As a donor of hydride and base to Lewis acids like borates and boranes [8]
- For deprotonating carbonyl compounds and amines to form enolates and amides respectively [8]
- In cyclization-condensation, rearrangement, elimination, and reducing reactions [8]
Is it Safe
KH is a pyrophoric compound and thus ignites in contact with air. It also produces flammable gases upon reacting with water [1]. Exposure to potassium hydride may cause eye damage and skin corrosion or burns [1].
- References
- Potassium Hydride – Pubchem.ncbi.nlm.nih.gov
- Potassium Hydride in Paraffin: A Useful Base for Organic Synthesis – Pubs.acs.org
- Potassium Hydride – Chemspider.com
- Potassium Hydride – Americanelements.com
- Science and Celebrity: Humphry Davy’s Rising Star – Sciencehistory.org
- Potassium Hydride And Sodium Hydride – Web.stanford.edu
- Potassium Hydride – Webelements.com
- Potassium Hydride – Onlinelibrary.wiley.com
- PKa Values of Common Bases – Uwindsor.ca