Nucleophilic Aromatic Substitution
What is Nucleophilic Aromatic Substitution?
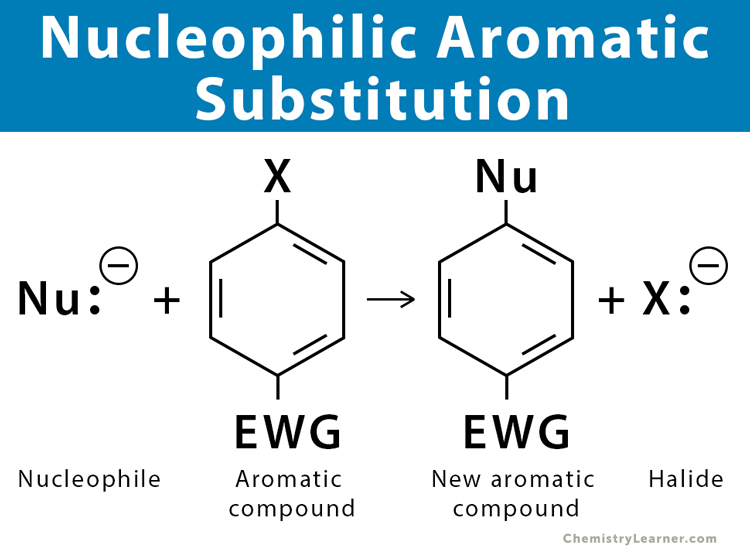
Nucleophilic aromatic substitution is a nucleophilic substitution reaction in which a nucleophile displaces a leaving group bonded to the carbon ring of an aromatic compound. In this reaction, a nucleophile attacks an aromatic ring that is deficient in electrons. The presence of an electron-withdrawing group facilitates the reaction. A typical leaving group is a halide, like fluoride or chloride [1-6].
Conditions for Nucleophilic Aromatic Substitution
Here are the facts and conditions for nucleophilic aromatic substitution reaction.
- The attacking species must be a strong nucleophile, i.e., it must have electrons that can bond with the carbon atom of the ring.
- The aromatic ring is poor in electrons, i.e., electron-deficient. The presence of an electron-withdrawing group activates the ring and makes it electron deficient.
- The leaving group is usually a nucleophilic species like a halide (X–) attached to a carbon atom.
- The nucleophile attacks at a site determined by the leaving group’s position, not by steric and electronic factors.
Examples of Nucleophilic Aromatic Substitution
Here is an example of nucleophilic aromatic substitution [2-6].
1-chloro-4-nitrobenzene (Cl-(C6H4)-NO2) reacts with sodium methoxide (CH3ONa) to produce 1-methoxy-4-nitrobenzene (CH3O-(C6H4)-NO2).
Cl-(C6H4)-NO2 + CH3ONa → CH3O-(C6H4)-NO2 + NaCl
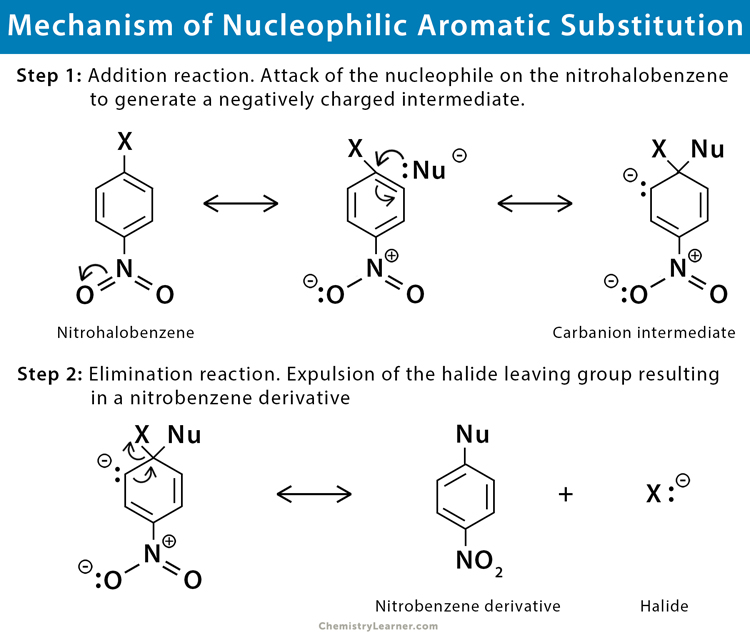
Mechanism of Nucleophilic Aromatic Substitution
There are four main mechanisms for the nucleophilic aromatic substitution involving a benzene ring, nucleophile (Nu), and a leaving group (LG) [2-7]. However, the addition-elimination mechanism is very common.
This mechanism is similar to electrophilic substitution, except that an anion instead of a cation intermediate is involved. The introduction of an electron withdrawing group, like nitro (NO2), makes the system vulnerable to nucleophilic attacks on the aromatic ring. Therefore, the nucleophile adds to the aromatic ring to form a delocalized anion, eliminating the halide leaving group.
FAQ
Ans. Radical-nucleophilic aromatic substitution is a type of nucleophilic substitution reaction in which the intermediate is a free radical species.