Nickel
What is Nickel
Nickel (pronunciation NICK-‘l [2]), represented by the chemical symbol or formula Ni [1], is a hard, ductile and malleable element belonging to the family of transition metals [3, 5]. Naturally occurring Ni is a mixture of 5 stable isotopes with mass numbers 58, 60, 61, 62 and 64 [1, 3]. Besides that, it has 23 synthetic isotopes with known half-lives [3]. It doesn’t react with water and oxygen (air) under normal and ambient conditions respectively [6].
Where Is It Found
Though the metal is occasionally found free in nature, it is principally found in its ores that are chiefly pyrrhotite and pentlandite (nickel-iron sulfides), millerite and garnierite. It is extracted from its minerals by roasting and reduction methods [1, 3]. A large amount of nickel reached the earth through meteorites that struck it [1].
History
Origin of Its Name: The name is the short form of the German ‘kupfernickel’ meaning either St. Nicholas’s copper or devil’s copper [1].
Who Discovered Nickel: Swedish chemist, Axel Fredrik Cronstedt [1, 2].
When Was It Discovered: Its discovery year is 1751 [1].
How Was It Discovered
Cronstedt, while working in Stockholm in 1751, investigated a new mineral (that is now called nickeline) that was found in a mine at Los, Hälsingland in Sweden. He presumed that it contained copper but what he extracted was a new metal that he named and announced as nickel in 1754. However, many chemists were of the opinion that it was an alloy of arsenic, cobalt, copper and iron though, in reality, they were present only as trace impurities. Pure nickel could be produced and confirmed of its elemental nature only in 1775 by another Swedish chemist, Torbern Bergman [1].
Nickel Identification |
|||
| Atomic Number | 28 [1] | ||
| CAS Number | 7440-02-0 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 10 [1] | 4 [1] | d [1] | |
Properties and Characteristics of Nickel
General Properties |
||
| Atomic mass | 58.693 atomic mass units [1] | |
| Atomic weight | 58.693 [1] | |
| Mass number | 58 [7] | |
| Molar mass/molecular weight | 58.693 g/mol [8] | |
Physical Properties |
||
| Color/appearance | Silvery-white [3] | |
| Luster | Yes [7] | |
| Melting point/freezing point | 1455°C, 2651°F [1] | |
| Boiling point | 2913°C, 5275°F [1] | |
| Density | 8.90 g cm-3 [1] | |
| State of matter at room temperature (normal phase) | Solid [1] | |
| Thermal Conductivity | 90 W/(m K) [9] | |
| Electrical Conductivity | 1.43X107 S/m [10] | |
| Specific heat capacity | 0.440 J/g oC [11] | |
| Specific gravity | 8.9 [12] | |
| Resistivity | 6.99X10-8 ohm-m [10] | |
| Hardness (Mohs scale) | 638 MPa [13] | |
| Yield strength (annealed) | 110 MPa [14] | |
Chemical Properties |
||
| Oxidation state/Oxidation number | -1, +1, +2, +3, +4 [1] | |
| Flammability | Yes (Ni powder) [15] | |
Magnetic Properties |
||
| Magnetic ordering | Ferromagnetic [4] | |
Atomic Data of Nickel (Element 28)
| Valence electrons | 10 [17] | |||||||
| Quantum numbers | ||||||||
| – n | 3 [16] | |||||||
| – ℓ | 2 [16] | |||||||
| – m ℓ | 0 [16] | |||||||
| – m s | -½ [16] | |||||||
| Electron configuration (noble gas configuration) | [Ar] 3d84s2 [1] | |||||||
| Crystal structure | Cubic face-centered [17] | |||||||
| Atomic structure | ||||||||
| – Number of Electrons | 28 [3] | |||||||
| – Number of Neutrons | 30 [3] | |||||||
| – Number of Protons | 28 [3] | |||||||
| Energy levels [3] | ||||||||
| – First Energy Level | 2 | |||||||
| – Second Energy Level | 8 | |||||||
| – Third Energy Level | 16 | |||||||
| – Fourth Energy Level | 2 | |||||||
| Radius of atom | ||||||||
| – Atomic Radius | 1.97 Å [1] | |||||||
| – Covalent Radius | 1.17 Å [1] | |||||||
| Electronegativity (Pauling scale) | 1.91 [1] | |||||||
| Ionization energy
(kJmol-1) [1] |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 737.129 | 1753.027 | 3395.32 | 5297 | 7338.67 | 10420 | 12833 | 15631 | |
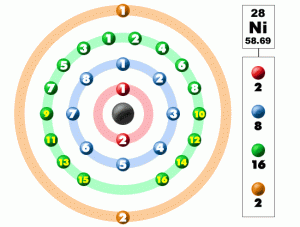
Nickel Electron Configuration (Bohr Model)
Nickel Uses
- In making stainless steel and other alloys, alkaline batteries and coins [15].
- It is used in electroplating and as a catalyst [15].
- Making electrical appliances such as electric ovens and toasters [1].
- Nickel is added to glass to give it a green color [1].
Is It Dangerous
Nickel is carcinogenic. It may cause skin and asthma-like allergies. When inhaled or brought in contact, it may adversely affect skin, eyes, nose and lungs [3, 15]. Serious health conditions such as cardiovascular diseases, neurological disorders, etc. are also caused by nickel poisoning [18]. However, as a trace mineral required by the human body, it has a number of health benefits [19]. Foods high in nickel are soya beans, chocolate, almonds, nuts and legumes [20].
Interesting Facts
- The metal does not rust and is corrosion resistant even when red hot [1, 3].
Nickel (Ni Element) Cost
The pure metal is priced at $7.7 for every 100 gram and in bulk, the same quantity costs $1.9 [3].
- References
- http://www.rsc.org/periodic-table/element/28/nickel
- https://education.jlab.org/itselemental/ele028.html
- https://www.chemicool.com/elements/nickel.html
- http://info.ee.surrey.ac.uk/Workshop/advice/coils/mu/
- https://www.lenntech.com/periodic/elements/ni.htm
- http://objetoseducacionais2.mec.gov.br/bitstream/handle/mec/10496/reactions/28.htm
- http://www.elementalmatter.info/mass-numbers.htm
- https://www.webqc.org/molecular-weight-of-Ni.html
- https://www.engineeringtoolbox.com/thermal-conductivity-metals-d_858.html
- https://www.thoughtco.com/table-of-electrical-resistivity-conductivity-608499
- http://www2.ucdsb.on.ca/tiss/stretton/database/specific_heat_capacity_table.html
- https://www.reade.com/reade-resources/reference-educational/reade-reference-chart-particle-property-briefings/25-specific-gravity-table-metals-minerals-ceramics-substance
- http://www.prvky.com/elements/nickel.html
- http://www.substech.com/dokuwiki/doku.php?id=commercially_pure_nickel_270_high_purity_nickel
- https://nj.gov/health/eoh/rtkweb/documents/fs/1341.pdf
- http://chemistry-reference.com/q_elements.asp?Symbol=Ni
- https://environmentalchemistry.com/yogi/periodic/Ni.html
- https://www.environmentalpollutioncenters.org/nickel/
- https://www.webmd.com/vitamins/ai/ingredientmono-1223/nickel
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3667300/










