Mitsunobu Reaction
Definition: What is Mitsunobu Reaction?
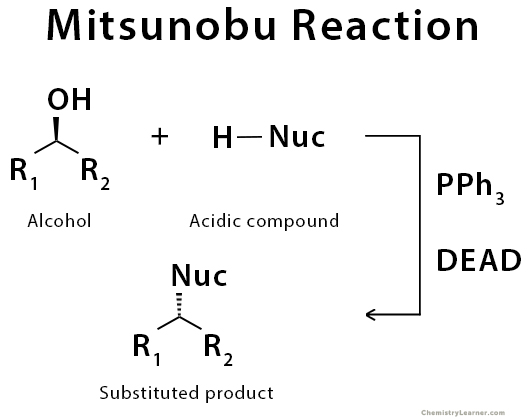
The Mitsunobu reaction is a common SN2 reaction taking advantage of phosphorous chemistry. It is an organic reaction that converts primary or secondary alcohol into a variety of functional groups (e.g., ester) using triphenylphosphine and an azodicarboxylate such as diethyl azodicarboxylate (DEAD) or diisopropyl azodicarboxylate (DIAD). The alcohol reacts with the phosphine to create an excellent leaving group then undergoes an inversion of stereochemistry in a standard SN2 reaction as the nucleophile displaces it. The Mitsunobu reaction proceeds under mild, mostly neutral conditions, and typically at 0 °C to room temperature [1-6].
The history of this reaction goes back to 1967 when Japanese Oyo Mitsunobu reported it.
Examples of Mitsunobu Reaction
The chiral alcohol, 2-octanol, reacts with benzoic acid with complete Walden-inversion, to give the corresponding ester [1].
Mechanism of Mitsunobu Reaction
The triphenylphosphine combines with DEAD to generate a phosphonium intermediate that binds to the alcohol oxygen, activating it as a leaving group. Substitution by the carboxylate, mercaptyl, or other nucleophile completes the process [4-6].
Intramolecular Mitsunobu Reaction
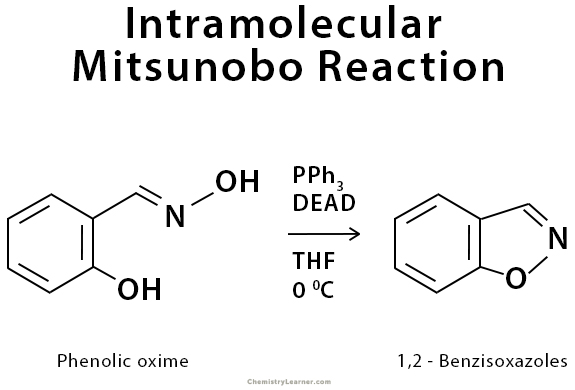
Intramolecular Mitsunobu reaction leads to cyclic products. The intramolecular attack of activated oximes by phenols is used in the synthesis of 1,2-benzisoxazoles. 2-Ureido phenols and 2-ureido anilines undergo intramolecular Mitsunobu reactions to deliver N2-substituted benzoxazoles and benzimidazoles, respectively [7].
Applications of Mitsunobu Reaction
The Mitsunobu reaction has found widespread use in many fields because of its high reliability and extensive versatility. It is mostly used in the total synthesis of biologically active natural products [8].
References
- Definition, examples, and mechanism – Tcichemicals.com
- Definition – Userpage.fu-berlin.de
- Definition – Slideshare.net
- Definition and mechanism – Chemistry.mcmaster.ca
- Definition and mechanism – Organic-chemistry.org
- Definition and mechanism – Name-reaction.com
- Intramolecular Mitsunobu reaction – Pubs.rsc.org
- Applications – Onlinelibrary.wiley.com