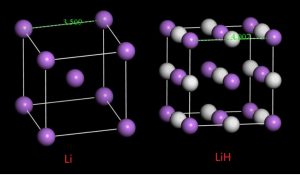
Lithium Hydride
Lithium hydride, represented by the chemical formula LiH, is a crystalline or powdery inorganic compound [1]. The alkali metal hydride has a molecular mass less than 8 and is the lightest ionic compound [1, 2].
How to make Lithium Hydride
LiH can be synthesized by the reaction of solid lithium with hydrogen gas, which is represented by the following chemical equation [5]:
2Li + H2 → 2LiH
The reaction occurs rapidly at temperatures over 600 °C, and the yield is increased to 98% [5]. It can also occur at 29 °C, but the yield is much lower [5].
Lithium hydride is also produced by combining lithium aluminum hydride and sodium hydroxide through metathesis in tetrahydrofuran (THF) [6]:
LiAlH4 + NaH → NaAlH4 + LiH
Reactions with Other Compounds
LiH reacts with air to produce Li2O, LiOH, and Li2CO3, while it forms nitrogenous compounds in moist air [5]. It spontaneously undergoes a chemical reaction with water which is given as follows [5]:
LiH + H2O → Li+ + H2 + OH–
Lithium hydride reacts with sulfur dioxide to form lithium dithionite at a temperature above 50 °C [5]:
2LiH + 2SO2 → Li2S2O4 + H2
When it reacts with acetylene, it produces hydrogen and lithium carbide [5]:
LiH + LiOH →Li2O + H2
Properties and Characteristics of Lithium Hydride
General Properties |
|
| Molar Mass/Molecular Weight | 7.948 g/mol [1] |
Physical Properties |
|
| Color and Appearance | Off-white, gray, or translucent; light bluish-gray lumps in commercial product [1] |
| Odor | Odorless [1] |
| Melting Point | 680 °C, 1256 °F [1] |
| Boiling Point | 900-1000 °C, 1650-1830 °F (decomposes) [1] |
| Density | 0.76-0.8 g cm-3 [1] |
| State of matter at room temperature | Solid [1, 4] |
| Solubility | Somewhat soluble in dimethylformamide; it reacts with diethyl ether, ammonia, and ethanol |
| Solubility in Water | Reacts [1] |
| Magnetic Susceptibility (χ) | -4.6 X 10-6 cm3/mol |
| Heat Capacity (C) | 3.51 J/(g.K) |
Atomic Properties |
|
| Crystal Structure | Octahedral cubic (NaCl-type) [7] |
What is It Used for
- Synthesis of hydrogen because LiH has the highest H2 content of any hydride [4]
- Manufacturing catalysts used for polyolefin plastics [8]
- Missiles, aircraft, and rocket fuel [8]
- Degasification, deoxidization, and desulfurization in the metallurgical industry [8]
- Forming alloys with magnesium and aluminum for aerospace use [8]
- Shielding and absorbing neutrons in mobile nuclear reactors, used along with lithium-7 [9]
Is It Toxic
Repeated contact or overexposure can cause acute irritation of skin, mucous membranes, and eyes [1]. When burned, it forms irritating alkali fumes [1]. LiH forms dust clouds that may explode when they come in contact with heat, flame, or oxidizing materials [1].
- References
- Lithium Hydride – Pubchem.ncbi.nlm.nih.gov
- Lithium Hydride – Chem.libretexts.org
- Lithium Hydride – Chemspider.com
- Lithium Hydride – Americanelements.com
- Technical Memorandum Compilation Of The Properties Of Lithium Hydride – Ntrs.nasa.gov
- Lithium Aluminium Hydride – Cs.Mcgill.ca
- Lithium Hydride – Webelements.com
- Lithium Hydride – Toxnet.nlm.nih.gov
- Lithium Hydride – A Space Age Shielding Material – Ntrs.nasa.gov