Heat of Combustion
The heat of combustion is the heat released by a substance when it undergoes complete combustion in the presence of oxygen at standard temperature and pressure conditions. When the amount is quantified for one mole of the substance, we call it molar heat of combustion [1-4].
How to Calculate Heat of Combustion
The heat of combustion is calculated by using a bomb calorimeter. A stoichiometric mixture of fuel and oxidizer is kept in a steel container at 25 °C and initiated by an ignition device. The reaction is then allowed to complete. Hydrogen and oxygen react during combustion, producing water vapor. The vessel and its contents are then cooled to the original 25 °C. The heat of combustion is determined as the heat is released [1-3].
Formula
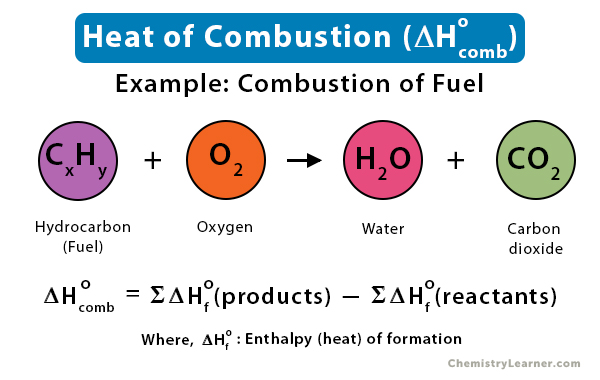
Theoretically, the heat of combustion is calculated from the difference between the heats of formation (ΔHfo) of the products and the reactants under standard conditions [1-3].
ΔHcombo = ΔHfo (products) – ΔHfo (reactants)
Symbol: ΔHcombo
Unit: Joules/mole (J/mol), Joules/gram (J/g), or J/m3
Standard conditions refer to a temperature of 25 ˚C and pressure of 105 Pa.
Example
When fuels containing hydrocarbons are combusted in oxygen, the resulting products are carbon dioxide and water. A large amount of energy is released in the process. Let us look at a few examples [1-3].
1. Methanol (CH3OH)
Methanol combusts in the presence of oxygen, releasing 726 kJ/mol of energy.
CH3OH (l) + O2 (g) → CO2 (g) + 2 H2O (l) ΔHcombo = – 726 kJ/mol
2. Propane (C3H8)
Propane combusts in the presence of oxygen, releasing 2220 kJ/mol of energy.
C3H8 (g) + 5 O2 (g) → 3 CO2 (g) + 4 H2O (l) ΔHcombo = – 2220 kJ/mol
Heating Values of Combustion
The heat of combustion for fuels is expressed as the HHV (Higher Heating Value) or LHV (Lower Heating Value) [1-3].
Lower Heating Value assumes that the water formed during a combustion process is in the vapor state after completing the process. Therefore, the energy required to vaporize water is not realized as heat.
Higher Heating Value assumes that all the water in a combustion process is in a liquid state after completion. The enthalpy change for the reaction assumes a common temperature of the substances before and after combustion.
The following table gives the molar heat of combustion or HHV for a few common substances [5].
| Substance | State | Formula | Molar Heat of Combustion, ΔHcombo (kJ/mol) |
|---|---|---|---|
| Hydrogen | gas | H2 | 286 |
| Methane | gas | CH4 | 891 |
| Ethane | gas | C2H6 | 1561 |
| Propane | gas | C3H8 | 2220 |
| Butane | gas | C4H10 | 2878 |
| Pentane | liquid | C5H10 | 3509 |
| Methanol | liquid | CH3OH | 726 |
| Ethanol | liquid | C2H5OH | 1367 |
| 1-Propanol | liquid | C3H7OH | 2021 |
| 1-Butanol | liquid | C4H9OH | 2676 |
| Acetone | liquid | CH3(CO)CH3 | 1790 |
| Benzene | liquid | C6H6 | 3268 |
| Toluene | liquid | C6H5CH3 | 3910 |
| Carbon (graphite) | solid | C | 394 |
Solved Problems
Problem 1: What is the molar heat of combustion of liquid ethanol (C2H5OH)?
Solution
The combustion reaction is
C2H5OH (l) + 3 O2 (g) → 2 CO2 (g) + 3 H2O (l)
We use the following formula:
ΔHcombo = ΔHfo (products) – ΔHfo (reactants)
=> ΔHcombo = [(2 mol) (ΔHfo (CO2)) + (3 mol) (ΔHfo (H2O))] – [(1 mol) ( ΔHfo (C2H5OH)) + (3 mol) (ΔHfo (O2))]
Refer to our article on enthalpy of formation for the ΔHfo values.
ΔHfo (CO2) (g) = -393.5 kJ/mol, ΔHfo (H2O) (l) = -285.8 kJ/mol, ΔHfo (C2H5OH) (l) = -277.6 kJ/mol, (ΔHfo (O2) (g) = 0
Therefore,
ΔHcombo = [(2 mol) (-393.5 kJ/mol) + (3 mol) (-285.8 kJ/mol)] – [(1 mol) (-277.6 kJ/mol) + (3 mol) (0)] = -1366.8 kJ/mol
Let us convert this into kJ/g.
-1366.8 kJ/mol =-1366.8 kJ/46.1 g = -29.6 kJ/g