Dieckmann Condensation
Definition: What is Dieckmann Condensation?
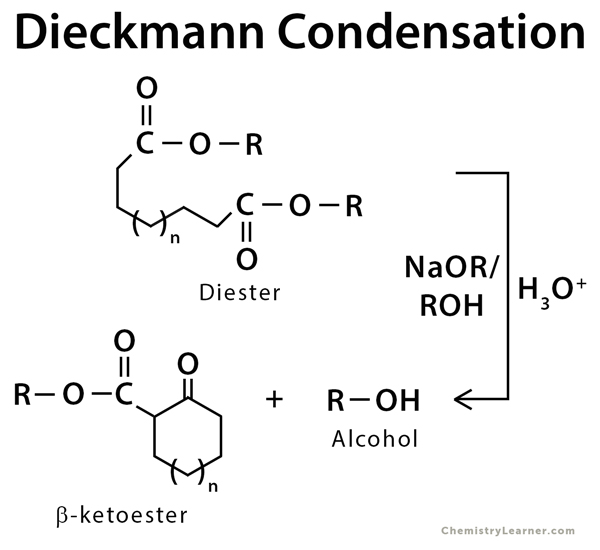
The Dieckmann condensation is an organic reaction which is used to form a carbon-carbon bond between two tethered ester groups using an alkoxide base in alcohol. The resulting product is a cyclic β-ketoester. This reaction is an intermolecular form of Claisen condensation [1-3].
The history of Dieckmann condensation goes back to 1894 when German chemist Walter Dieckmann first reported it in a journal.
Examples of Dieckmann Condensation
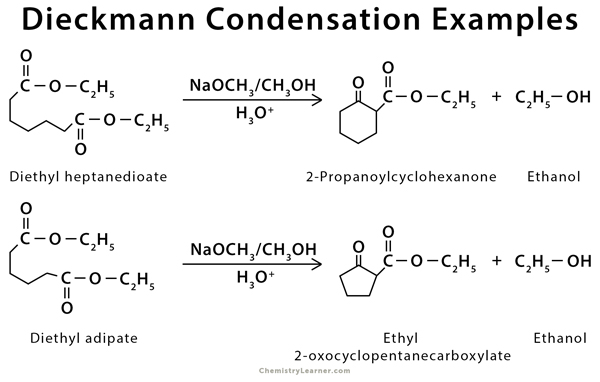
Examples of Dieckmann condensation include conversion of diethyl adipate into ethyl 2-oxocyclopentanecarboxylate and diethyl heptanedioate (pimelate) into 2-propanoylcyclohexanone [4-5].
Mechanism of Dieckmann Condensation
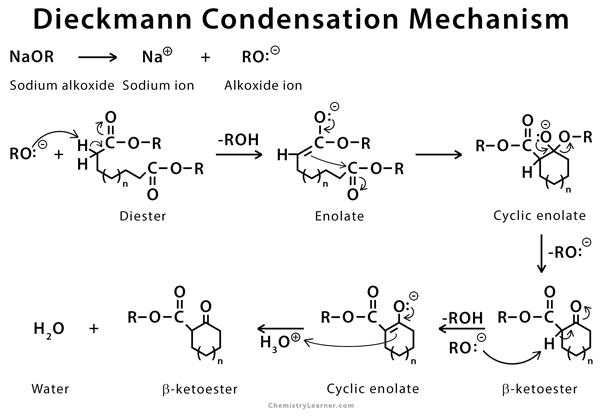
The mechanism is similar to that of Claisen condensation. A strong base removes the alpha proton resulting in an enolate. The enolate then attacks the carbonyl carbon of another ester molecule, resulting in a cyclic β-ketoester and regenerating the conjugate base. The base then deprotonates the cyclic compounds, and an acid workup is required to produce the final cyclic β-ketoester [6-8].
Applications of Dieckmann Condensation
Dieckmann condensation is used in the preparation of cyclic indole. There has been a report of preparing five-membered pyrroles through this condensation method [9].
References
- Definition – Name-reaction.com
- Definition – Organic-chemistry.org
- Definition – Chem.ucalgary.ca
- Example – Chem.libretexts.org
- Example – Chtf.stuba.sk
- Mechanism – Ochempal.org
- Mechanism – Chem.ucla.edu
- Mechanism – Synarchive.com
- Application – Conservancy.umn.edu