Coordination Number
The coordination number in a complex molecule or crystal is the number of atoms, ions, or molecules bonded to the central atom. The ion or molecule surrounding the central atom is called a ligand. The coordination number is also called ligancy. Another way to define coordination number is the number of attachment points between a metal and a ligand. Coordination complexes are essential for transitional metals, although some main group elements also form them [1-4].
Factors Affecting Coordination Number
The coordination number can vary from as low as 2 to as high as 16. Several factors determine its value [1].
- Shape and size of ligands – The bigger the ligand, the smaller the coordination number.
- Size of metal ion – The larger the metal ion, the more ligands can fit around it. The opposite is also true. Large metals and small ligands lead to high coordination numbers. Small metals with large ligands lead to low coordination numbers.
- Electron configuration of metal ion – Only those electron configurations that stabilize the electron energies are preferred.
- Intermolecular interactions – The intermolecular interaction between neighboring complexes in the solid state and between the complexes and solvent molecules in solution leads to different coordination numbers.
How to Determine Coordination Number
The coordination number for different compounds can be calculated as follows [2,3]:
1. Molecule or polyatomic ion – Count the number of atoms bonded to the central atom. Note that only sigma bonds are counted. Pi bonds do not contribute to the coordination number.
Examples
a. Methane (CH4) – Carbon has a coordination number 4 since it has four hydrogen atoms bonded to it.
b. Ethene (C2H4) – Carbon has a coordination number 3 since each carbon is bonded to two hydrogen and one carbon atom.
c. Ethyne (C2H2) – Carbon has a coordination number 2 since each carbon is bonded to one hydrogen and one carbon atom.
d. Copper (I) chloride ion or [CuCl2]-1 – Copper (Cu) has a coordination number 2 since two chlorine (Cl) atoms surround it.
e. Triiodomercurate or [HgI3]-1 – Mercury (Hg) has a coordination number 3 since three iodine (I) atoms are bonded to it.
2. Crystal – Count the number of neighboring atoms that extend in all directions.
Examples
a. Diamond (C) – Carbon has a coordination number 4 since each carbon is placed at the center of a regular tetrahedron formed by four other carbon atoms.
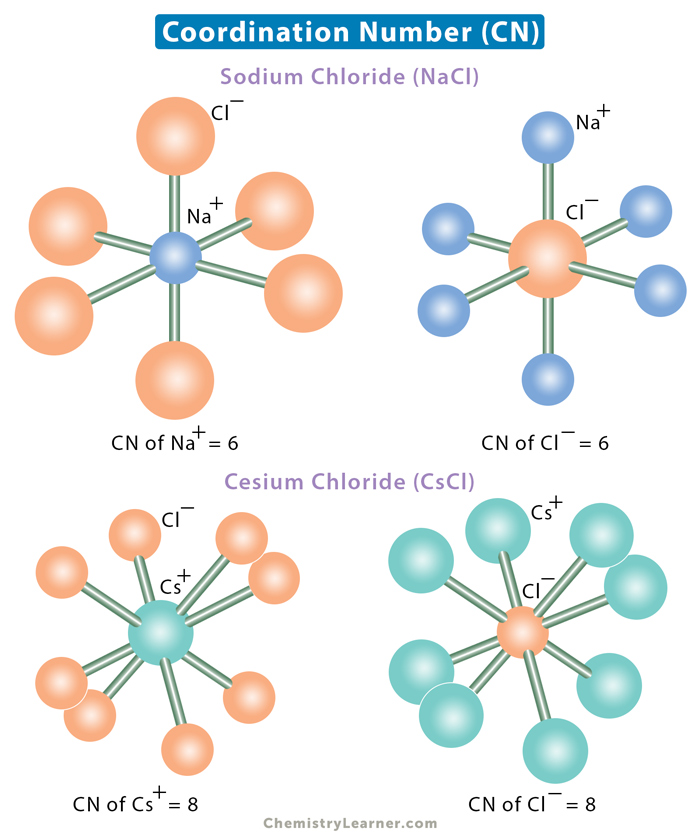
b. Sodium chloride (NaCl) – Six chloride ions (Cl–) closely surround a single sodium ion (Na+). Also, six Na+ ions immediately surround each Cl– ion. The coordination number of sodium and chlorine is 6.
c. Cesium chloride (CsCl) – The central cesium ion (Cs+) is surrounded by eight chloride ions. Each Cl– is also surrounded by eight Cs+. The coordination number of cesium and chlorine is 8.
d. Titanium (IV) oxide (TiO2) – Titanium ion (Ti4+) is surrounded by six oxide ion (O2-) ions. O2- ions are surrounded by three Ti4+ ions. The coordination numbers of Ti4+ cation and O2- anion are 6 and 3, respectively. It also explains why the titanium (IV) oxide formula is TiO2.
3. Complex ion or coordination complex – Count the ligands attached to the central atom.
Examples
a. Nickel tetracarbonyl or Ni(CO)4 – Nickel (Ni) has a coordination number 4 since four CO ligands are attached to the Ni atom.
b. Hexachloroplatinate or [PtCl6]2– – The coordination number of platinum (Pt) is 6 since it is connected to six Cl– ligands.
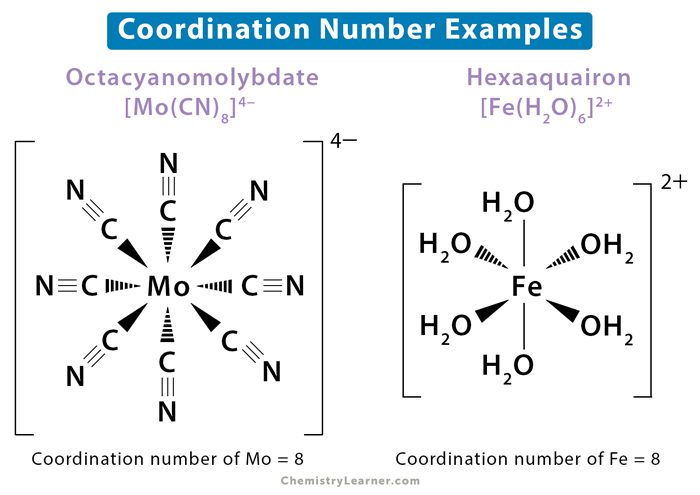
c. Hexaaquairon or [Fe(H2O)6]2+ – The coordination number of iron (Fe) is 6 since it is connected to six water (H2O) ligands.
d. Octacyanomolybdate or [Mo(CN)8]4− – The coordination number of molybdenum (Mo) is 8 since there are eight cyanide (CN–) ligands attached to it.
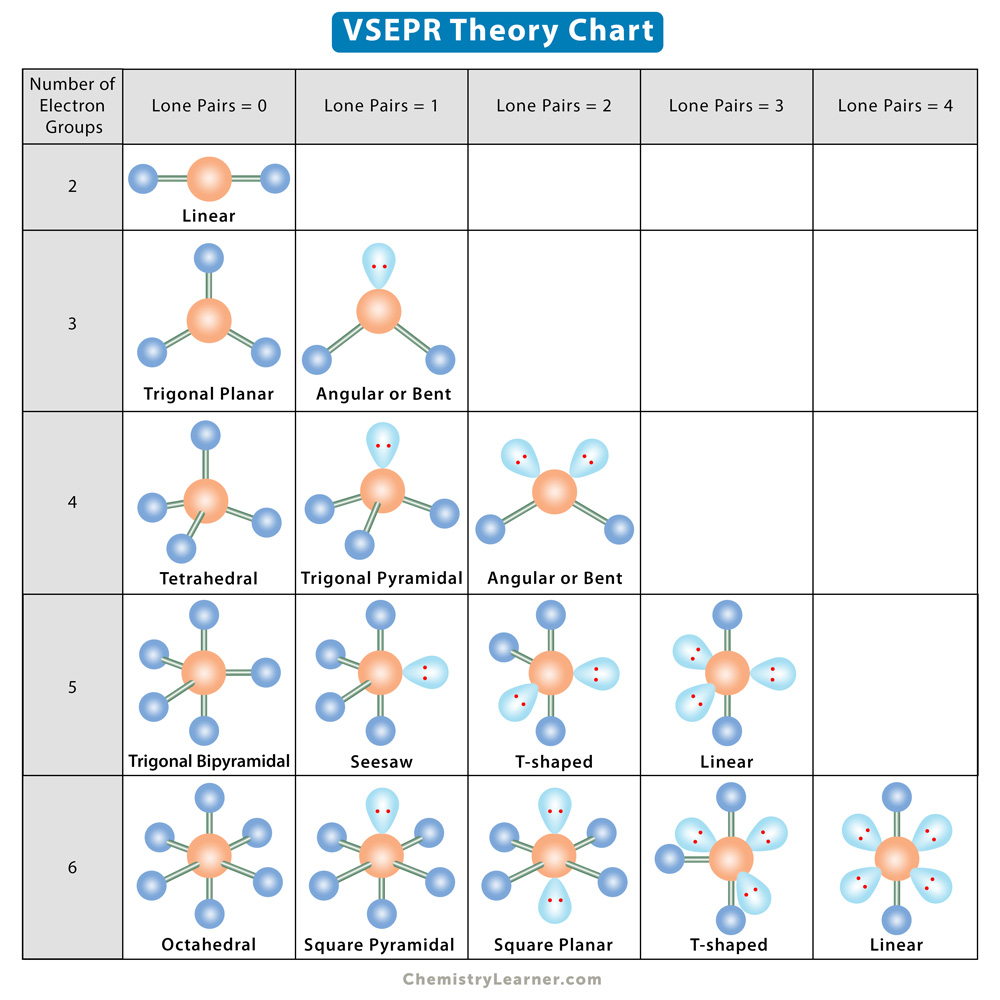
Coordination Number and Molecular Geometry
The coordination number of the central metal atom is used to determine the structure of the molecules. All the possible geometries are listed in the table below [2].
| Coordination Number | Geometry |
|---|---|
| 2 | Linear |
| 3 | Trigonal planar, trigonal pyramidal, or T-shaped |
| 4 | Tetrahedral or square planar |
| 5 | Square pyramidal or trigonal bipyramidal |
| 6 | Hexagonal planar, trigonal prism, or octahedral |
| 7 | Capped octahedron, capped trigonal prism, or pentagonal bipyramidal |
| 8 | Cubic, hexagonal bipyramidal, square antiprism, or dodecahedron |
| 9 | Three-faced centered trigonal prism |
| 10 | Bicapped square antiprism |
| 11 | All-faced capped trigonal prism |
| 12 | Cuboctahedron |