Clemmensen Reduction
Definition: What is Clemmensen Reduction?
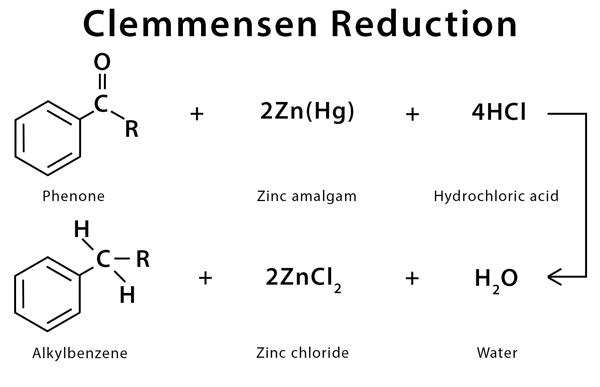
Clemmensen reduction is a chemical reaction used to reduce an aldehyde or ketone to an alkane using zinc amalgam and concentrated hydrochloric acid. The process is carried out by replacing the double bond between carbon and oxygen by two hydrogen atoms. The zinc dissolves during the process, and hence, this reaction can be classified as dissolving metal reduction. Clemmensen reduction generally applies to aromatic ketones, like aryl-alkyl ketones. It is not so much applied to non-aromatic ketones [1-3].
The history of this reaction goes back to 1913 when Danish chemist Erik Christian Clemmensen discovered.
Examples of Clemmensen Reduction
Clemmensen reduction can be used to reduce acetophenone and benzaldehyde to ethylbenzene and methylbenzene, respectively [4, 5].
Mechanism of Clemmensen Reduction
The mechanism of this reaction is not studied widely, which is a limitation of Clemmensen reduction. The reduction may take place through two possible mechanisms. The first one is called the carbanionic mechanism, where the zinc attacks the protonated carbonyl directly. The second one is called the carbenoid mechanism, which is a radical process. The reduction happens on the surface of the zinc metal. In this article, the carbanionic mechanism is shown [6-8].
FAQs
Ans. Clemmensen reduction is made under acidic conditions, whereas the Wolff-Kishner reaction takes place under basic conditions.
References
- Definition – Organic-chemistry.org
- Definition – com
- Definition – ucla.edu
- Example – libretexts.org
- Example – com
- Mechanism – Name-reaction.com
- Mechanism – com
- Mechanism – Researchgate.net
- Definition – Organic-chemistry.org
- Definition – com
- Definition – ucla.edu
- Example – libretexts.org
- Example – com
- Mechanism – Name-reaction.com
- Mechanism – com
- Mechanism – Researchgate.net