Home / Organic Chemistry / Beckmann Rearrangement
Beckmann Rearrangement
Table Of Contents
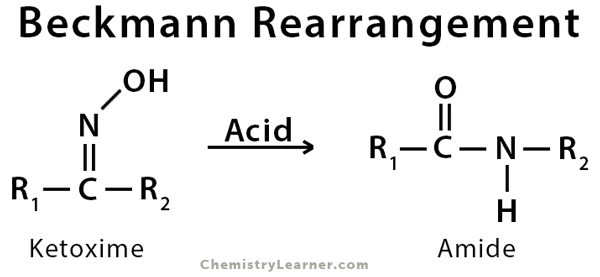
Definition: What is Beckmann Rearrangement?
Beckmann rearrangement is an acid-catalyzed reaction of ketoximes into amides. The yield of this reaction increases at elevated temperatures. The reaction is named after German pharmacist and chemist Ernst Otto Beckmann [1-2].
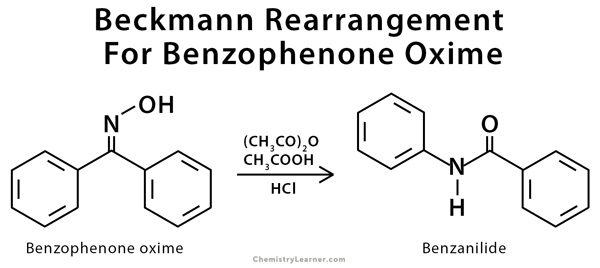
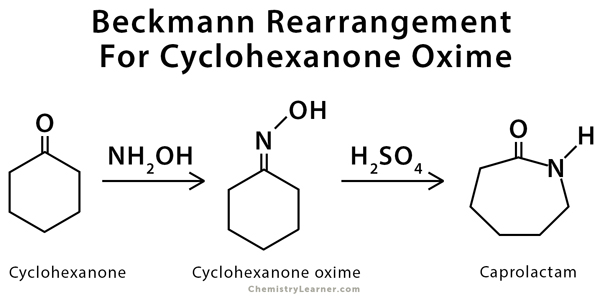
Examples of Beckmann Rearrangement
Beckmann rearrangement is used in the conversions of benzophenone oxime to benzanilide and cyclohexanone oxime to caprolactam [3].
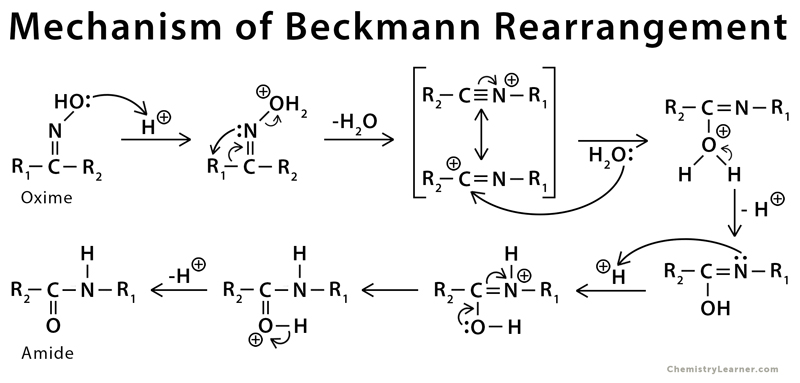
Mechanism of Beckmann Rearrangement
The mechanism of the Beckmann rearrangement follows the same pattern as a pinacol reaction. The acid converts the oxime OH into a leaving group, and an alkyl group migrates on to the nitrogen as water departs. The product cation is then trapped by water to give an amide [4-8].
Applications of Beckmann Rearrangement
- In the synthesis of caprolactam, which is used in the production of nylon 6
- For the synthesis of paracetamol by converting a ketone to a ketoxime with the help of hydroxylamine
- In the synthesis of various steroids and drugs
- In the production of some of the chloro bicyclic lactams
References
- Definition and example – Chem.libretexts.org
- Definition – Researchgate.net
- Example – Pubs.acs.org
- Mechanism – Adichemistry.com
- Mechanism – Chemtube3d.com
- Mechanism – Synarchive.com
- Mechanism – Name-reaction.com
- Mechanism – Nature.com