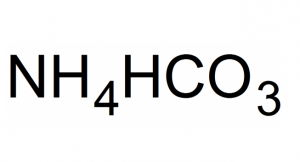Ammonium Bicarbonate
Ammonium bicarbonate, commonly as baking soda, ammonium hydrogen carbonate, and powdered baking ammonia, is a colorless inorganic compound represented by the chemical formula (NH4)HCO3 or CH5NO3 [1, 2]. In IUPAC nomenclature, it is named azanium hydrogen carbonate [1]. It decomposes readily into water, ammonia, and carbon dioxide [3].
Ammonium Bicarbonate Composition and Synthesis
Its preparation involves the reaction of carbon dioxide and ammonia in a cold condition, with ammonium bicarbonate forming as a solid white precipitate. It is represented by the following equation [4]:
NH3 + CO2+ H2O → (NH4)HCO3
It was once commercially produced by dry distillation of organic matters like leather, horn, and hair. Also called ammonium sesquicarbonate, the material also produces ammonium carbonate ((NH4)2CO3) and ammonium carbamate (NH4CO2NH2).
Properties and Characteristics of Ammonium Bicarbonate
General Properties |
|
| Molar Mass/Molecular Weight | 79.055 g/mol [1] |
Physical Properties |
|
| Color and Appearance | White or colorless [1] |
| Odor | Faint ammoniacal [8] |
| Melting Point | 41.9 °C, 107.4 °F (decomposes) [1] |
| Density | 1.586 g cm-3 [1] |
| pH | 7.8 [5] |
| State of matter at room temperature | Crystalline solid [1] |
| Solubility | Soluble in water, insoluble in acetone, benzene, methanol, and ethanol [1] |
| Solubility in Water | 17.4 g/100 cc at 20 °C [1] |
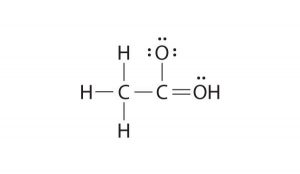
Ammonium Bicarbonate Lewis Structure with Covalent Bonding
Uses
- As a leavening agent for baked goods like crackers, cookies, and cream-puff doughs in the food industry [1].
- Producing ceramics, porous plastics, dyes, and pigments [1].
- For scale removal in boilers and as a foaming agent for rubber [1]. Removal of gypsum from heat exchangers and processing equipment [1].
- Added to compost heaps for accelerating the rate of decomposition and using them as fertilizers [1].
- It is a good buffer (because of its volatile nature) in pharmaceutical applications like lyophilization (freeze-drying) for producing injectable and oral dosage forms [5].
- Used as a component of human and vet medications [1].
Is It Safe
Accidental ingestion of large amounts of ammonium bicarbonate may be harmful and cause serious damage to the health resulting in diarrhea, systemic poisoning, and increased urine production [6]. Its inhalation can cause respiratory irritation with the body’s response to the irritation causing further lung damage [1, 6]. Its contact with skin is also known to cause irritation [1].
- References
- Ammonium Bicarbonate – Pubchem.ncbi.nlm.nih.gov
- Ammonium Bicarbonate – Chemspider.com
- Ammonium Bicarbonate – Sigmaaldrich.com
- Carbon Dioxide React with Ammonium Hydroxide – Chemiday.com
- Ammonium bicarbonate (A6141) – Product Information – Sigmaaldrich.com
- Ammonium Bicarbonate Material Safety Data Sheet – Datasheets.scbt.com