Aluminum Sulfide
Aluminum sulfide, also called dialuminium trisulfide, is an ionic compound of aluminum and sulfur represented by the chemical formula Al2S3 [1, 2]. It exists in different crystalline forms and hydrolyzes to aluminum hydroxide/oxide [3, 4].
Aluminum Sulfide Identification |
|
| CAS Number | 1302-81-4 [1, 2] |
| PubChem CID | 16684788 [6] |
| ChemSpider ID | 140154 [1] |
| EC Number | 215-109-0 [1, 2] |
| InChI Key | ZPXDOGRYMSWBBM-UHFFFAOYSA-N [6] |
How is Aluminum Sulfide Prepared
Its synthesis through direct heating of aluminum and sulfur can be represented by the following equation [7]:
2Al + 3S → Al2S3
However, the reaction is highly exothermic, and it is not desirable to heat the entire mixture [7]. Also, sulfur is a volatile reactant that melts away before the reaction takes place [7]. Therefore, lead sulfide is often used for the preparation of Al2S3 [7]:
2Al + 3PbS → Al2S3 + 3Pb
Reactions with Other Compounds
Aluminum Sulfide and Water
When aluminum sulfide is exposed to moisture in the atmosphere, it undergoes hydrolysis reaction to form aluminum hydroxide and hydrogen sulfide [5].
Al2S3 + 6H2O → 2Al(OH)3 + 3H2S
Aluminum Sulfide and Hydrochloric Acid
Aluminum sulfide upon reacting with hydrochloric acid forms hydrogen sulfide and aluminum chloride, as shown below [8]:
Al2S3 + 6HCl → 2AlCl3 + 3H2S
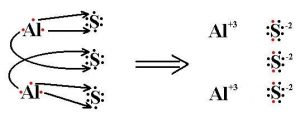
Aluminum Sulfide Lewis Structure
Properties and Characteristics of Aluminum Sulfide
General Properties |
|
| Molar Mass/Molecular Weight | 150.158 g/mol [1] |
Physical Properties |
|
| Color and Appearance | Gray powder or granules [2] |
| Melting Point | 1,100 °C, 2,012 °F [2] |
| Boiling Point | 1,500 °C, 2,732 °F [2] |
| Density | 2.32 g cm-3 [2] |
| State of matter at room temperature (solid/liquid/gas) | Solid [2] |
| Solubility | Insoluble in (CH3)2CO (acetone) [9] |
| Solubility in Water | Reacts and decomposes in H2O [9] |
| Heat Capacity (C) | 105.1 J/mol K |
| Standard Molar Enthalpy | -724 kJ/mol [9] |
| Standard Molar Entropy | 116.9 J/mol K |
Atomic Properties |
|
| Crystal Structure | Trigonal, tetragonal, hexagonal [3, 4] |
What is It Used for
- Producing organic compounds like ethanethiol and chemicals required for tanning and making paper [10]
- Manufacturing cathodes used in lithium-sulfur solid-state batteries [11]
- Preparing hydrogen sulfide that has several industrial uses [11]
Is Aluminum Sulfide Toxic
Exposure to aluminum sulfide can cause skin and severe eye irritation [12]. Inhalation of dust, vapor, or fumes can cause difficulty in breathing [12].
- References
- Aluminum Sulfide (2:3) – Chemspider.com
- Aluminum Sulfide – Americanelements.com
- Synthesis and Crystal Structure of a Novel Hexagonal Modification of Al2S3 with Five-coordinated Aluminum – Onlinelibrary.wiley.com
- High-pressure Spinel Type Al2S3 and MnAl2S4 – Adsabs.harvard.edu
- Aluminum Sulfide React with Water – Chemiday.com
- ZPXDOGRYMSWBBM-UHFFFAOYSA-N – Pubchem.ncbi.nlm.nih.gov
- Preparation of Aluminum Sulfide – Prepchem.com
- Aluminum Sulfide React with Hydrogen – Chemiday.com
- Aluminum Sulfide [Al2S3] – Chembk.com
- What Are the Uses of Aluminum Sulfide? – Reference.com
- Aluminum Sulfide – Chemicalbook.com
- Safety Data Sheet Aluminum Sulfide – Ltschem.com