Acetanilide
- Acetanilide Identification
- Acetanilide Chemical Formula
- Acetanilide Structure
- Preparation of Acetanilide
- Acetanilide Physical and Chemical Properties
- Acetanilide Uses
- Acetanilide in Pharmaceuticals
- Bromination of Acetanilide
- Nitration of Acetanilide
- Acetanilide Material Safety Data Sheet (MSDS)
- Acetanilide Availability
Acetanilide is a white solid organic compound mainly used in organic synthesis. Other names of this compound include N-phenylacetamide, acetanilide and acetanil. It was formerly used for treating fever and headache and was known by its brand name Antifebrin.
Acetanilide Identification
CAS number: 103-84-4
ChemSpider: 880
UNII: SP86R356CC
EC-number: 203-150-7
KEGG: C07565
ChEBI: CHEBI: 28884
ChEMBL: CHEMBL269644
Acetanilide Chemical Formula
The chemical formula for this compound is C6H5NH(COCH3).
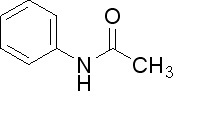
Acetanilide Structure
This is the structure of one Acetanilide molecule:
Picture 1 – Acetanilide
Preparation of Acetanilide
This organic compound can be synthesized by the reaction of acetic anhydride with aniline. It happens in the following way:
C6H5NH2 + (CH3CO)2O → C6H5NHCOCH3 + CH3COOH
This production method of Acetanilide used to be a traditional experiment in introductory lab classes of organic chemistry. However, it was replaced by the preparation process of other compounds (aspirin or paracetamol) to avoid using aniline which is a suspected carcinogen.
Acetanilide Physical and Chemical Properties
Here are some of the basic properties of this organic compound:
Appearance: It is a white solid with a flaky appearance.
Odor: This is an odorless compound.
Solubility: Acetanilide is a little soluble in water. It is also soluble in diethyl ether, ethanol, benzene and acetone.
Molecular Weight: Its molecular weight is 135.17 g/mol.ility
Melting Point: It has a melting point of 114.3 °C.
Boiling Point: The boiling point for this substance is 304 °C.
Density: The density of Acetanilide is 1.219 g/ cm3.
Specific Gravity: It has a specific gravity of 1.214.
Vapor Pressure: The vapor pressure for this compound is 2 Pa at 20 °C.
Acetanilide Uses
The uses of this organic material have decreased over the past few years due to its toxicity. However, it is still used for some industrial purposes:
- It has application as an inhibitor in hydrogen peroxide.
- This compound is also used for stabilizing cellulose ester varnishes.
- It is applied in the intermediation in accelerator synthesis of rubber.
- This organic compound is used for manufacturing 4-acetamidobenzenesulfonyl chloride which is one of the key ingredients for producing sulfa drugs.
- This compound is used as a precursor for synthesizing pharmaceuticals like penicillin.
- It was experimentally used as a photographic developer in the 19th century.
Acetanilide in Pharmaceuticals
This aniline derivative was the first one found to have both analgesic and antipyretic properties. A. Cahn and P. Hepp introduced this derivative into medical practice under the brand name of Antifebrin in 1886. However, the evident toxicity of this substance promoted the need of searching for less toxic derivatives of aniline like phenacetin. In 1948, it was discovered that Acetanilide metabolizes into paracetamol in human body and this resulting paracetamol had the analgesic and antipyretic properties. As result, Acetanilide was slowly replaced by its metabolite paracetamol which is still quite well known in the pharmaceutical field.
Bromination of Acetanilide
A chemical reaction in which a Bromine atom (Br) is introduced into another molecule is called Bromination. Acetanilide can be brominated by reacting it with Bromine atoms. Following is the equation for this chemical reaction:
KBrO3 + 3 C6H5NH(COCH3) + 3 HBr → 3 Br ̶ C6H5NH(COCH3) + 3 H2O + KBr
The reaction between Potassium Bromate (KBrO3), Acetanilide and Hydrogen Bromide (HBr) brominates the Acetanilide and produces water (H2O) and Potassium Bromide.
Nitration of Acetanilide
A nitro group substitutes a hydrogen atom in the aromatic ring of Acetanilide in this electrophilic aromatic substitution reaction. In this reaction, the aromatic ring functions as the nucleophile while the nitronium ion functions as the electrophile. It can be considered as a reaction of a Lewis base with a Lewis acid.
Acetanilide Material Safety Data Sheet (MSDS)
It is a toxic material that can be combustible at high temperatures. This compound can cause serious health hazards for humans if comes into direct contact.
Toxicology
Direct skin and eye contact will result in severe irritation. It can cause skin allergies and irritation of the respiratory tract. More serious effects may include blood abnormalities. This organic powder targets different human organs including blood, central nervous system, kidney and skin.
First Aid Measures
Eye Contact: Any contact lenses should be removed immediately in case of accidental eye contact. One should flush the eyes with plenty of water at least for 15 minutes. Immediate medical aid is required.
Skin Contact: Victim should wash the contaminated skin area with disinfectant soap and water before applying an emollient lotion to it. It is important to get immediate medical assistance.
Inhalation: The victim should be removed to fresh air in case of accidental inhalation of this powder. Oxygen and artificial respiration should be provided in case the victim is experiencing breathing problems. One should immediately seek medical attention.
Ingestion: It is not advisable to induce vomiting without proper direction from a qualified physician. Any tight clothing like tie, collar and waistband should be loosened. One should never give anything by mouth if the victim is unconscious. Immediate medical assistance is important.
Personal Safety Measure
One should use NIOSH approved splash goggles, lab coats, protective gloves and dust respirator for proper personal protection while handling this material.
Fire Fighting Measures
It can be combustible at very high temperatures. Fire fighters should use proper fire fighting gear when extinguishing a fire caused by or around this powder. Dry chemical powder can be used to put out a small fire while foam or water spray should be used for extinguishing a large fire. One should never use water jet for this purpose.
Storage and Disposal
This powdered organic compound should be stored in tightly sealed containers and kept in a cool and well ventilated area. Empty containers pose a risk of causing fire hazards so they should be treated under a fume hood in order to evaporate any residue in them.
Acetanilide Availability
This organic powder is available at online stores at reasonable prices.
Acetanilide is used for numerous industrial purposes around the world. Although it has lost many of its uses since it was discovered to be a toxic material, it is still quite useful in many fields.
- References





I want to ask about the particle size of acetanilide for my final assigment. Do you know how much is the particle size of acetanilide which is sold in the market? Thank you
The method of preparation of different types of compounds is satisfactory. …..