Ionic Bond
What is an Ionic Bond?
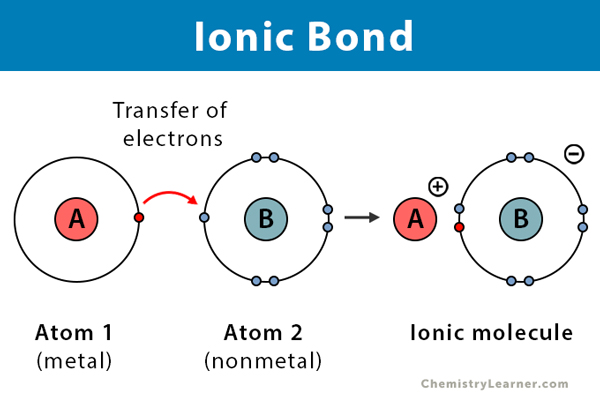
An ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between oppositely charged ions in a compound or molecule.
Ionic bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom following the octet rule. The atom that loses electrons becomes a positively charged ion, known as a cation. The atom that gains electrons becomes a negatively charged ion, known as an anion. When the two ions combine via ionic bond, they form ionic compounds.
The types of elements forming ionic bonds are metals and nonmetals [1-4].
How do Ionic Bonds Form
Electronegativity is a property of an atom, measuring how strongly it attracts or holds onto electrons. Ionic bonds are formed when there is a high electronegativity difference between the atoms. The high difference in electronegativities results in an electrostatic attraction between the electrons of one atom and the other atom’s nuclei. When atoms transfer electrons, they achieve a stable, inert gas electronic configuration. As a result, the outermost (valence) shells of the atoms are complete [1-4].
Properties and Characteristics of Ionic Bonds
Ionic bonds affect the properties of ionic compounds, mostly the physical properties [3,4]. They are mentioned below:
- Involves the transfer of electrons from one atom to another
- Formed between a metal and nonmetal with sharp electronegativity difference so that the nonmetal can attract the electrons from the metal
- The attraction between the nuclei and electrons is electrostatic with charge separation. They are the most reactive of all the bonds in the appropriate medium.
- Strength is directly proportional to the quantity of the charges and inversely proportional to the distance between the charged ions.
- Ionic compounds have high melting and boiling points. The atoms are held together in a lattice structure, and much energy is needed to melt the compounds.
- Ionic compounds generally have a lattice structure and are crystalline at room temperature.
- Ionic compounds are brittle, and they break along the planes where the pressure is applied.
- The ionic compound dissolves in water and other polar solvents very smoothly. The crystals break up into ions, which are the carriers of electricity. Aside, ionic compounds also conduct electricity in a molten state.
- Ionic compounds are insoluble in nonpolar solvents.
- Ionic compounds display a high rate of reaction.
- Bonds are non-directional, and so, ionic compounds do not exhibit isomerism.
Examples of Ionic Bond
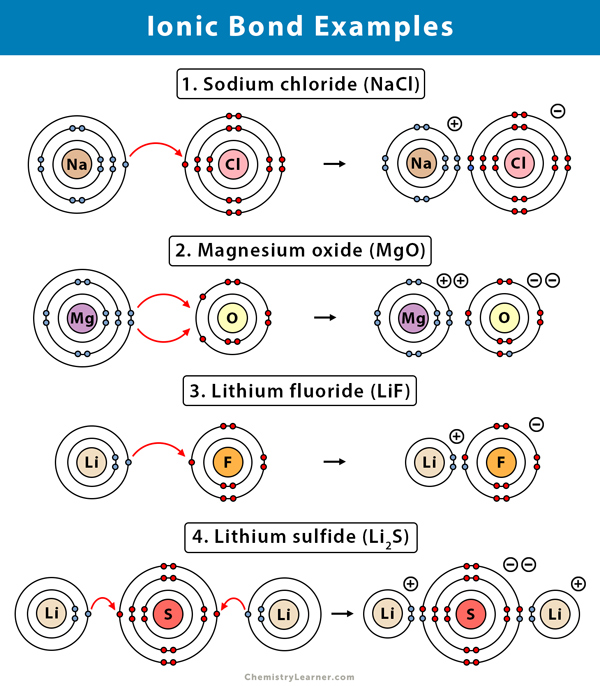
Here are some examples of ionic bonds along with their formulae [1-4].
1. Sodium Chloride (NaCl)
Sodium (Na) has a valency of one. It has only one electron in its outermost (valence) shell. Chlorine has seven electrons in its outermost shell and requires one electron to complete the shell. Therefore, sodium will donate its lone electron and become sodium ion (Na+), thereby taking the electronic configuration of its nearest inert gas, neon. Chlorine is more electronegative than sodium and will accept the electron to form a chloride (Cl–) ion. In this process, chlorine achieves its nearest inert gas configuration, i.e., argon. The two atoms combine to form an ionic bond, resulting in NaCl, which is commonly known as table salt.
2. Magnesium Oxide (MgO)
Magnesium (Mg) has two electrons in its outermost shell, and oxygen (O) has six. Magnesium will donate the two electrons to achieve its nearest inert gas configuration, i.e., neon. As a result, magnesium will become a magnesium ion (Mg2+). Oxygen (O) is more electronegative than magnesium. It requires two electrons to complete its outermost shell and achieve its nearest inert gas configuration, neon. Therefore, it will attract the two electrons from magnesium and transform into an oxide ion (O2-). Finally, an ionic bond results between the two atoms, and we get MgO.
3. Calcium Chloride (CaCl2)
Calcium (Ca) has two valence electrons, and chlorine (Cl) has seven. The former will give off its two electrons to form a calcium ion (Ca+). Chlorine is more electronegative than calcium will attract those electrons. Hence, two chlorine atoms will combine with calcium. Each will attract one electron, resulting in a chloride (Cl–) ion. As a result, there will be two ionic bonds between calcium and chlorine. The resulting product, CaCl2, is commonly known as rock salt.
4. Potassium Oxide (K2O)
Potassium (K) has one electron in its outermost shell. Oxygen (O) has six electrons in its outermost shell and requires two to complete the shell. It is more electronegative than potassium, and so it will attract two electrons from two potassium atoms forming an oxide ion (O–). Thus, both the potassium will donate one electron to oxygen, resulting in two potassium ions (K+). As a result, the molecule potassium oxide will have two ionic bonds.
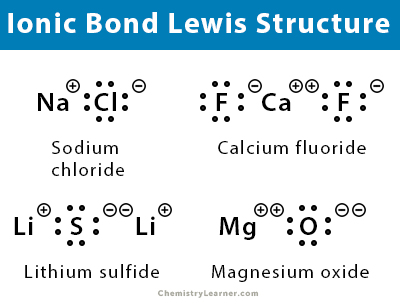
Ionic Bond Lewis Structure
Lewis dot structure can be used to represent compounds containing ionic bonds. In this method of representation, dots represent the outmost electrons around the atom.
FAQs
Ans. It depends upon the environment. In a vacuum, ionic bonds are stronger than covalent bonds because of the strong electrostatic interaction. However, in water, ionic bonds disassociate, making them weaker.
Ans. Sodium nitrate (NaNO3) is a compound with both ionic and covalent bonds.
Ans. No. Unlike covalent bonds, ionic bonds do not share electrons.
Ans. Hydrogen bonds are similar to ionic bonds in that they are formed through the attraction of atoms possessing opposite polarities.