Dipole-dipole Forces
Intermolecular interactions are essential for the stability of the molecule. There are several types of intermolecular forces, and dipole-dipole forces are one of them.
What are Dipole-dipole Forces
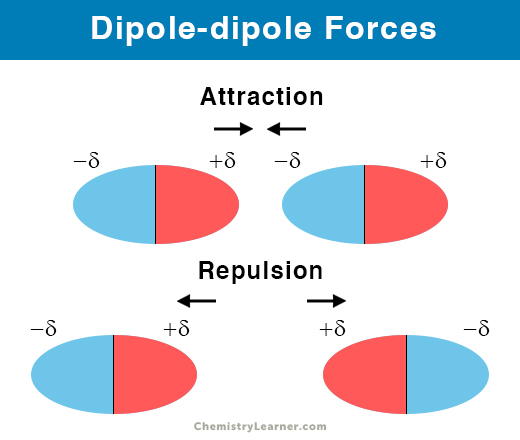
Dipole-dipole forces, also known as dipole-dipole interactions, are the electrostatic forces between two permanent polar molecules. Generally, the positive end of one molecule is attracted to the negative end of another molecule. As a result, the two molecules come closer, adding to the stability of the substance. This interaction is different from a regular ionic or covalent bond since there is no transfer or sharing of electrons.
What Causes a Dipole-dipole Interaction
Dipole-dipole interaction arises due to the uneven distribution of electrons in a molecule. The electrons congregate at one end of the molecule. Thus, the molecule acquires a partially negative charge at one end and a partial positive charge at the other end, thus making them polar. Two polar molecules with opposite charges will naturally attract one another.
Examples of Dipole-dipole Intermolecular Forces
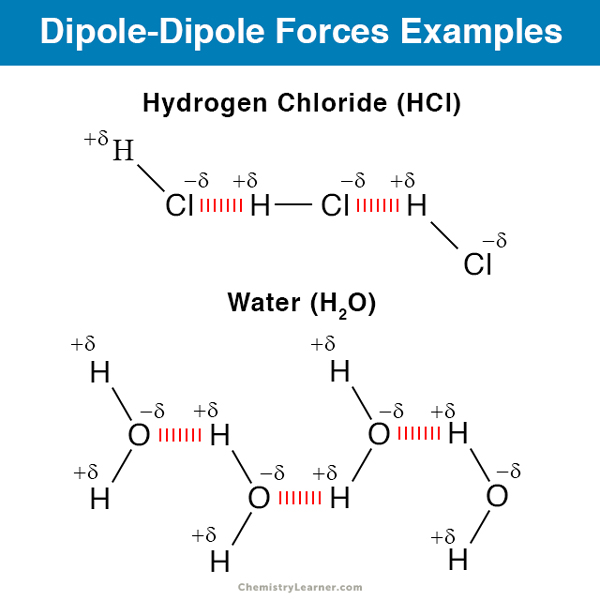
Examples of dipole-dipole forces include hydrogen chloride (HCl), hydrogen fluoride (HF), and water (H2O)
- Hydrogen chloride (HCl): HCl has a permanent dipole. The hydrogen atom has a partial positive charge, and the chlorine atom has a partially negative charge. When two HCl molecules are brought closer, the positive H of one molecule attracts the negative Cl of another molecule and forms a bond.
- Water (H2O): In H2O, two hydrogen (H) atoms are bonded to an oxygen (O) atom. As a result, the O-H bond acquires a permanent dipole. The hydrogen is partially positive, and the oxygen is partially negative. Therefore, H from one molecule will attract the O from another molecule resulting in a dipole-dipole force.
Properties
- Occurs between two permanent polar molecules
- The positive end attracts the negative end
- Have bond strength in the range of 5 to 20 kJ per molecule
- Weaker than ionic and covalent bonds
Applications
Dipole-dipole forces play an essential role in determining the properties of a substance. They are significant for the formation of stable compounds, including biological organisms. Aside, soft adhesives use dipole-dipole forces that allow them to stick on to surfaces. Some applications of the dipole-dipole interactions are:
Medical industry: Adhesives are used widely in medical devices, allowing a patch to stick to the skin. From wound dressings to transdermal patches, adhesives play a vital role in designing products for end-users.
Construction materials: Construction materials like sand, gravel, rocks, cement, and water have dipole-dipole forces that provide the desired strength. Aside, materials like plywood and plastics are often glued together using appropriate glues.
Electronic devices: Electronic components are joined together using soft adhesives. Besides, household gadgets consist of polar substances like diodes, metalloids, and semiconductors capable of inducing dipole.
Sports equipment: Material like carbon fiber is used to build sports equipment like racquets for tennis, badminton, and racquetball. These equipment are durable and require strong intermolecular forces of attraction.
Special Types of These Forces
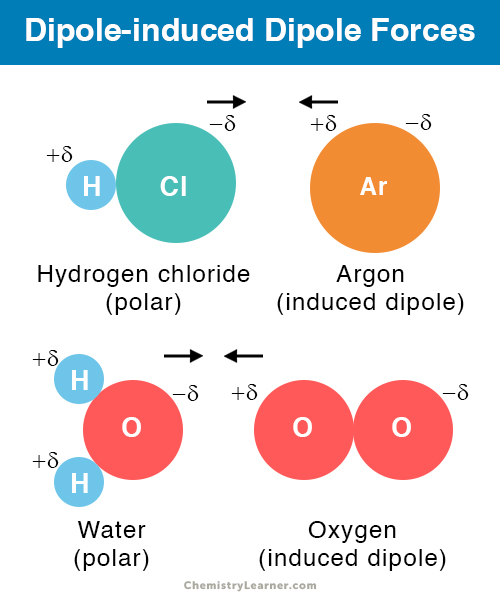
Dipole-induced Dipole Forces
A polar molecule can induce a dipole moment in a nonpolar molecule or an atom. When the two are brought together, the arrangement of electrons in the nonpolar molecule will be disrupted. As a result, it will acquire a partially positive charge and a partially negative charge. The two molecules will attract one another through dipole-induced dipole forces. It is a weaker interaction than the regular dipole-dipole interaction.
Examples
The interaction between hydrogen chloride (HCl) and argon (Ar); In this type of interaction, the positively-charge hydrogen (H) atom of one HCl molecule will attract the electrons from the Ar atom the negatively-charged Cl atom of another molecule will attract the Ar nucleus. As a result, a dipole moment will be induced in Ar.
Hydrogen Bonding
To compare hydrogen bonding with dipole-dipole forces, one must know the atoms present in a polar molecule. Hydrogen bonding is a special type of dipole-dipole interaction between the hydrogen atom of one polar molecule and an electronegative atom of another, like oxygen, chlorine, and fluorine. The hydrogen bond is essential in biology and biochemistry. It is responsible for maintaining the structure of the DNA molecule.
Examples
Hydrogen chloride (HCl), hydrogen fluoride (HF), and water (H2O).
FAQ
Ans. As Cl2 is not a polar molecule, it does not have dipole-dipole forces.
Ans. Dispersion forces are weaker than the dipole-dipole forces because they are caused by temporary dipoles. In contrast, dipole-dipole forces are caused by permanent dipoles.
Ans. London dispersion forces occur due to temporary dipoles, but dipole-dipole forces are attractive forces between molecules having permanent dipoles.
References
- Dipole-dipole Forces – Chem.purdue.edu
- Dipole-dipole Interactions – Chem.libretexts.org
- Dipole-dipole – Ch301.cm.utexas.edu
- Intermolecular Forces – Chemistry.msu.edu
- Intermolecular Forces – Chemistry.bd.psu.edu
- Intermolecular Forces – Chemistry.elmhurst.edu
- The Intermolecular Forces of Attraction – Chesquations.wordpress.com