Polar Covalent Bond
What is a Polar Covalent Bond?
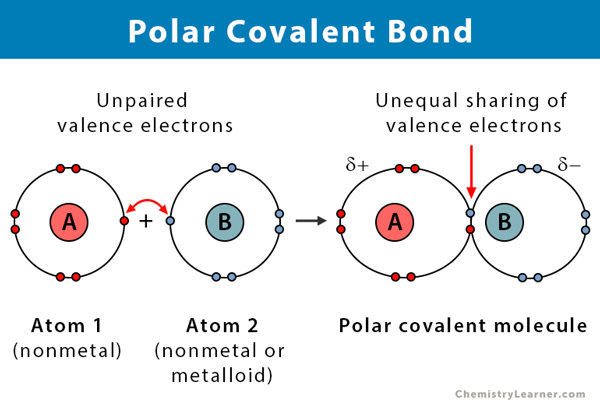
A covalent bond is formed when two atoms share electrons between them. The covalent bond is likely polar when the electrons are unequally shared. The skewness arises due to the difference in the electronegativities of the two atoms. Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. It is measured using a scale known as the Pauling scale and ranges from 0.7 to 4. The following table explains the type of chemical bond formed due to this electronegativity difference [1-10].
| Type of Chemical Bond | Electronegativity Difference |
|---|---|
| Nonpolar covalent | 0 |
| Slightly polar covalent | 0.1 to 0.4 |
| Polar covalent | 0.5 to 2 |
| Ionic | > 2 |
The more significant the difference in electronegativity, the greater the polarity and the bond strength. Hence, polar covalent bonds have an intermediate bond strength.
Properties of Polar Covalent Bonds
Here are some facts about polar covalent bonds.
- It takes place between two nonmetals or between a nonmetal and a metalloid
- Unequal sharing of electrons in a compound
- The electronegativity difference between atoms is between 0.1 and 2
- One end of the bond is positively charged, and the other end is negatively charged
- Polar compounds dissolve in a polar solvent
Examples of Polar Covalent Bonds
Here are some examples of a polar covalent bond with the electronegativity difference between the two bonding atoms [1-6]:
| Compound Name | Molecular formula | Bond | Electronegativity difference [1] |
|---|---|---|---|
Water | H2O | O-H | 1.4 |
| Hydrogen fluoride | HF | H-F | 1.9 |
| Hydrogen chloride | HCl | H-Cl | 0.9 |
Hydrogen bromide | HBr | H-Br | 0.7 |
Ammonia | NH3 | N-H | 0.9 |
Sulfur dioxide | SO2 | S=O | 1 |
| Hydrogen sulfide | H2S | H-S | 0.4 |
| Methanol | CH3OH | O-H | 1.4 |
An explanation of the polar covalent bond in some compounds is given below.
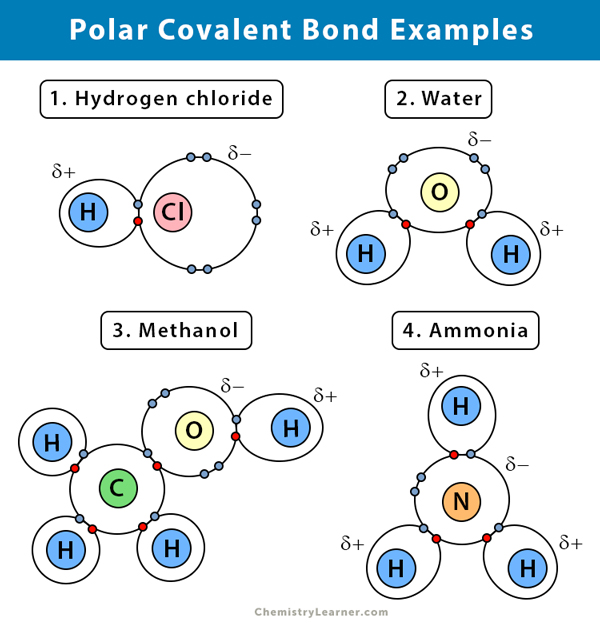
1. Water (H2O)
Water is a polar solvent. A polar covalent bond is created when the oxygen (O) atom, more electronegative than hydrogen, pulls the shared electrons towards itself. As a result, the oxygen atom has a partial negative charge associated with it. On the other hand, the hydrogens (H) are essentially protons and have a partial positive charge.
2. Hydrogen Chloride (HCl)
Hydrogen chloride is a polar covalent compound as the chlorine (Cl) atom is more electronegative than the hydrogen (H) atom. So, chlorine carries a partial negative charge, while hydrogen carries a partial positive charge.
3. Ammonia (NH3)
Ammonia is a polar molecule. The central atom, nitrogen (N), is bonded to three hydrogen (H) atoms. Since N is more electronegative than H, the electrons will tend to hang around the N atom.
4. Hydrogen Bromide (HBr)
The difference in electronegativity between hydrogen (H) and bromine (Br) is not too high. Therefore, the HBr gas has a covalent bond that is polarized. The hydrogen atom has a slight positive charge, and the bromine has a slight negative charge. As a result, the electron pair is attracted to the bromine.
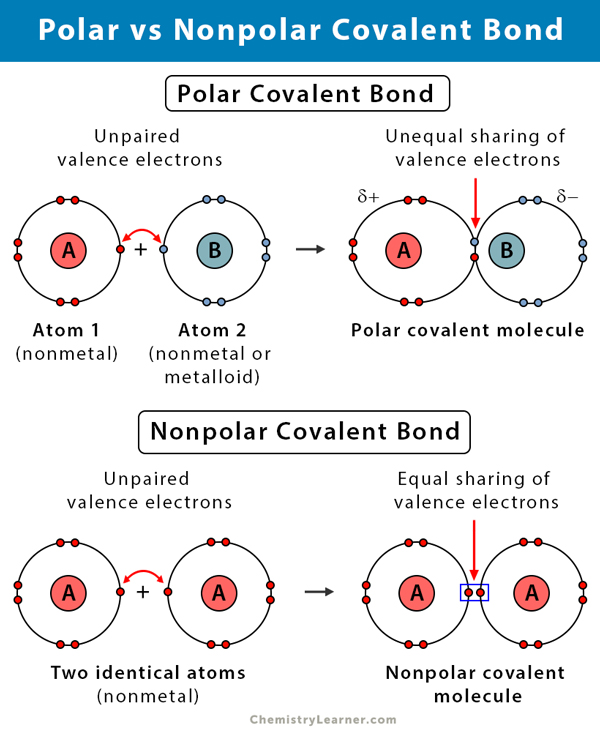
Difference Between Polar and Nonpolar Covalent Bonds [3]
The following table compares and contrasts polar and nonpolar covalent bonds.
Polar Covalent Bond vs. Nonpolar Covalent Bond | ||
| Polar | Nonpolar | |
|---|---|---|
Types of atoms | Between two nonmetals with different electronegativities | Between two nonmetals of same electronegativities |
|
Electronegativity difference |
0.1 – 2 |
0 |
|
Electron distribution |
Asymmetrical. Unequal sharing. |
Symmetrical. Equal sharing. |
|
Displacement of shared electrons |
Towards the more electronegative atom, making that part negative |
No displacement. Electrically neutral. |
|
Non-zero |
Zero | |
|
Presence of other types of bonds in compounds |
Van der Waals bond | |
|
Affinity towards water |
Hydrophilic |
Hydrophobic |
|
Physical properties of the compounds |
High melting and boiling points |
Low melting and boiling points |
|
Volatility of liquids |
Low volatility |
High volatility |
|
Solubility of compounds |
Soluble in polar solvents |
Soluble in nonpolar solvents |
|
Examples |
Water (H2O), ammonia (NH3), and hydrogen chloride (HCl) |
Hydrogen (H2), oxygen (O2), and nitrogen (N2) |