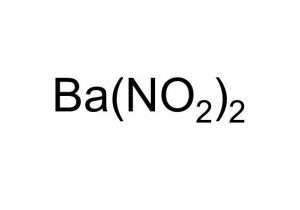Home / Inorganic Chemistry / Barium Nitrite
Barium Nitrite
Table Of Contents
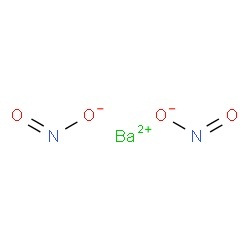
Barium nitrite represented by the chemical formula Ba(NO2)2 or BaN2O4 [2] that bears the IUPAC name barium dinitride [3] is a white crystalline powder that is soluble in water and alcohol [6]. It is a barium salt and an ionic compound [2].
Barium Nitrite Identification |
|
| CAS Number | 13465-94-6 [1] |
| PubChem CID | 166820 [2] |
| ChemSpider ID | 145952 [3] |
| EC Number | 236-709-9 [1] |
How to Make It
Barium nitrite can be prepared by decomposing silver nitrite by barium chloride [4].
2AgNO2 + BaCl2 = 2AgCl + Ba(NO2)2
It can also be made by pouring a warm solution of barium nitrate over lead and boiling the mixture for 1.5 to 2 hours [7].
Ba(NO3)2 + 2Pb = Ba(NO2)2 + 2PbO
Properties and Characteristics of Barium Nitrite
General Properties |
||
| Molar mass/molecular weight | 229.337 g/mol [2] | |
Physical Properties |
||
| Color/appearance | White crystals [1] | |
| Melting point/freezing point | 115°C, 239°F [1] | |
| Boiling point | N/A [1] | |
| Density | 3.173 g cm-3 [1] | |
| State of matter at room temperature (normal phase) | Solid [1] | |
Chemical Properties |
||
| Solubility in water | 630 g/l (at 20oC) [1] | |
| pH | >7 (basic) [5] | |
Atomic Properties |
||
| Crystal structure | Hexagonal [6] | |
Barium Nitrite Uses
- In research work in laboratories, in diazotization [1, 6].
- As a corrosion inhibitor [6].
- In making explosives [6].
Is It Dangerous
The compound can be highly toxic if swallowed or inhaled [2].
- References
- Barium Nitrite – Americanelements.com
- Barium Nitrite – Pubchem.ncbi.nlm.nih.gov
- Barium Nitrite – Chemspider.com
- Barium Nitrite, Ba(NO2)2 – Barium.atomistry.com
- Basic anions none think about why 1 anions of weak – Coursehero.com
- Barium Nitrite – Chemicalbook.com
- Preparation of barium nitrite – Prepchem.com