How to Read Periodic Table
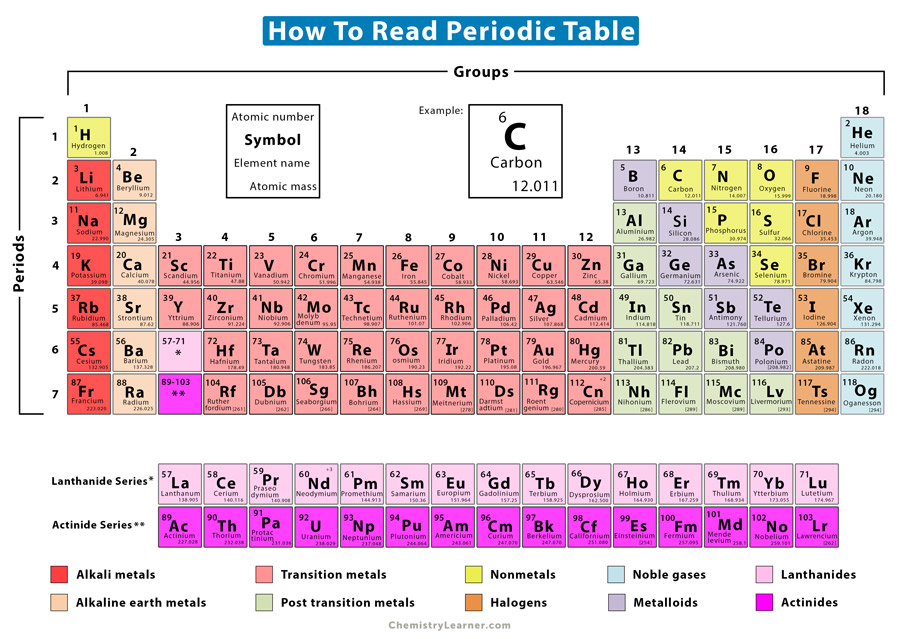
The periodic table is a helpful chart in chemistry as it provides the atomic properties of the elements. It consists of more than a hundred elements found naturally and artificially. The periodic table is divided into rows and columns. The rows are called periods, and there are seven of them. The columns are called groups, and there are 18 of them. The colors of the elements represent the different families [1-4].
How Are Elements Arranged Across a Period
Across a period, the elements are arranged according to their increasing atomic number. For example, let us consider the third period. The first element is sodium (Na), whose atomic number is 11. The last element in this period is argon (Ar), whose atomic number is 18. In between, there are elements whose atomic numbers range from 12 to 17 [1-10].
There are two rows of elements at the bottom of the periodic table. These are rare earth elements that are called lanthanides and actinides. Technically, they lie within Group 3 in the sixth and seventh periods, respectively.
How Are Elements Arranged Down a Column
An atom consists of a nucleus surrounded by electrons located in shells. Down a group, the elements are arranged according to their increasing number of electron shells or energy levels. Consider Group 17 as an example. The first element in this group is fluorine (F) which has two electron shells. Going down that group, the number of electron shells increases until we reach tennessine (Ts), which has seven shells [1-10].
Element Box
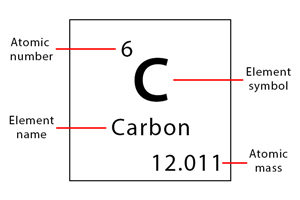
Let us now look at how elements are represented in the periodic table. Each element is shown in a square box with four critical pieces of information – atomic number, symbol, name, and atomic mass. All four parameters are essential to know the element’s atomic properties. These are discussed below [1-10].
Atomic Number: The atomic number is placed on the top of the box. It is the number of protons inside the nucleus or electrons in the various shells. Each neutral atom has the same number of protons and electrons.
Symbol: The elemental symbol consists of one or two letters and is placed below the atomic number. It represents the element’s name in abbreviated form. For example, oxygen is abbreviated as O. Not all symbols come from the element’s English name. Some of them are taken from their Latin names. For example, Ag is the elemental symbol of silver. It comes from silver’s Latin name, which is Argentum.
Name: The name of the element is placed under the symbol.
Atomic Mass: The atomic mass is placed at the bottom of the element box. It is the average of all the masses of the element’s isotopes, given that not all isotopes have the same abundance on Earth. It gives a rough estimate of the average number of all the protons and neutrons in the nucleus.
Example
To illustrate further, let us take the example of carbon (C). Its element box is shown below.
Carbon is located in Period 2, Group 14 of the periodic table. It is the lightest nonmetal. Let us study the information in the element box.
- Carbon has 6 protons and 6 electrons. Hence, its atomic number is 6.
- The elemental symbol of carbon is C, the first letter of its English name.
- The average atomic mass of carbon is 12.011. It is the average of all carbon isotopes. No single carbon isotope has a mass of 12.011 amu or atomic mass unit.