Formal Charge of Nitrate (NO3–)
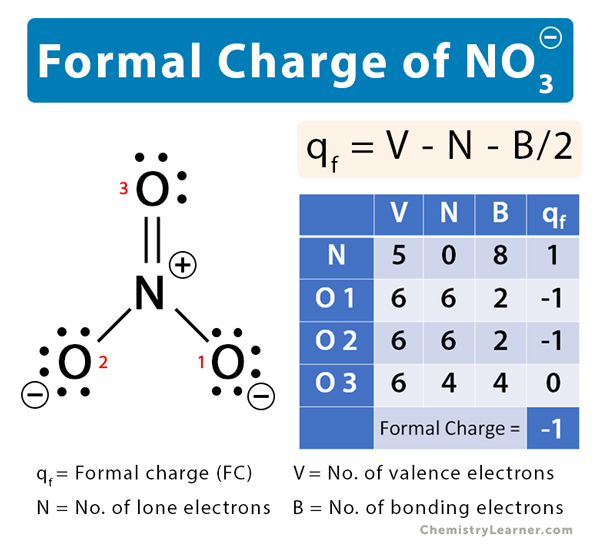
The nitrate ion (NO3–) consists of a nitrogen (N) atom with five valence electrons and three oxygen (O) atoms, each having six valence electrons. Nitrogen shares a double covalent bond with one oxygen atom (O=N+) and two single covalent bonds (O–-N+) with the remaining two oxygen atoms. Its molecular structure is shown below.
To find the formal charge of NO3–, let us calculate the formal charges on the atoms.
Nitrogen
V = 5, N = 0, B = 8
Therefore,
qf = 5 – 0 – 8/2 = 1
The formal charge on N in NO3– is 1.
Oxygen 1 and Oxygen 2
V = 6, N = 6, B = 2
Therefore,
qf = 6 – 6 – 2/2 = -1
The formal charge on two of the oxygen atoms in NO3– is -1.
Oxygen 3
V = 6, N = 4, B = 4
qf = 6 – 4 – 4/2 = 0
The net formal charge is: -1 + (2 x -1) + 0 = -1
The formal charge of NO3– is -1.