Home / Physical Chemistry / Methyl Ion (CH3–) Formal Charge
Methyl Ion (CH3–) Formal Charge
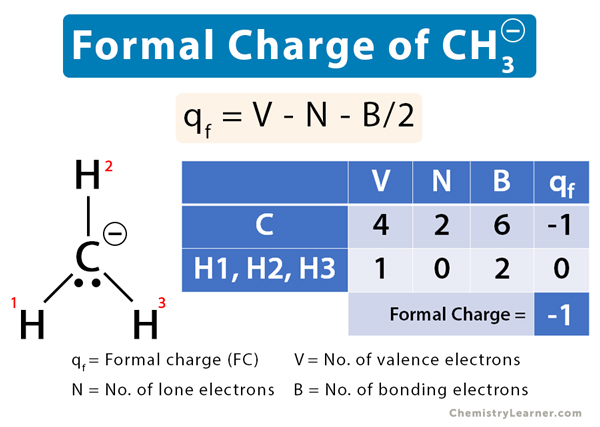
The methyl ion (CH3–) consists of a carbon (C) atom that is bonded to three hydrogen (H) atoms. Its molecular structure is shown below.
Let us calculate the formal charges on C and H and then determine the formal charge of CH3–.
Carbon
V = 4, N = 2, B = 6
Therefore,
qf = 4 – 2 – 6/2 = -1
Theformal charge on C in CH3– is -1.
All Three Hydrogen Atoms
V = 1, N = 0, B = 2
Therefore,
qf = 1 – 0 – 2/2 = 0
The net formal charge is: -1 + 3 x 0 = -1
The formal charge of CH3–is zero.