Home / Organic Chemistry / Fischer Indole Synthesis
Fischer Indole Synthesis
Table Of Contents
Definition: What is Fischer Indole Synthesis?
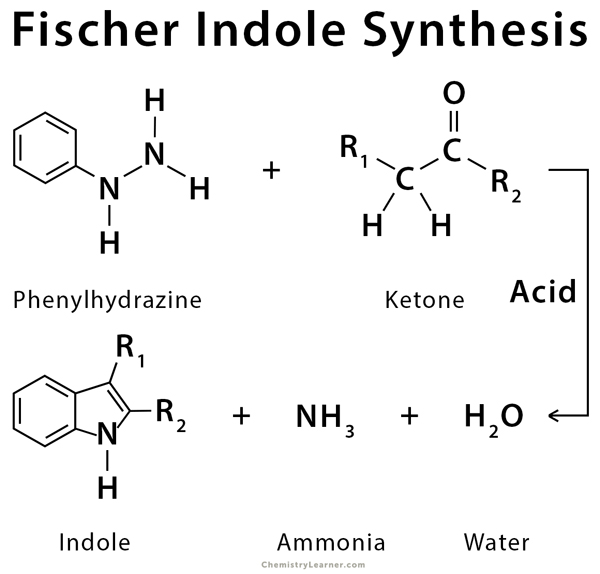
The Fischer indole synthesis is an organic reaction used to convert a phenylhydrazine and an aldehyde or ketone to an indole using an acid catalyst, like Brønsted or Lewis acids. An application of this reaction is the synthesis of antimigraine drugs belonging to the triptan class [1-8].
The history of this reaction goes back to 1883 when the reaction was first discovered by a German chemist Emil Fischer.
Examples of Fischer Indole Synthesis
The Fisher indole synthesis is used to synthesize 2-phenylindole and tetrahydrocarbazole [2,3,9].
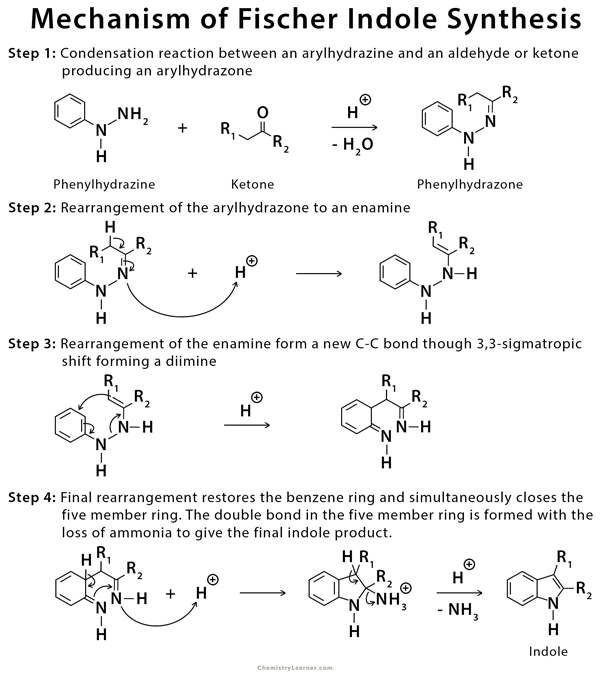
Mechanism of Fischer Indole Synthesis
Phenylhydrazone is formed from a condensation reaction of phenylhydrazine and an aldehyde or ketone. The phenylhydrazone converts into an indole in the presence of an acid catalyst [2-8,10].
References
- Definition – Chemtube3d.com
- Definition, example, and mechanism – Pubs.acs.org
- Definition, example, and mechanism – Researchgate.net
- Definition and mechanism – Name-reaction.com
- Definition and mechanism – Organic-chemistry.org
- Definition and mechanism – Ch.ic.ac.uk
- Definition and mechanism – Synarchive.com
- Definition and mechanism – Onlinelibrary.wiley.com
- Example – Sciencedirect.com
- Mechanism – Nature.com
- Mechanism – Users.miamioh.edu