Fries Rearrangement
Definition: What is Fries Rearrangement?
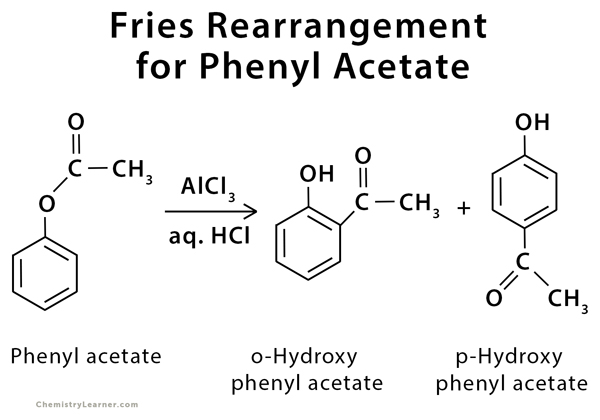
The Fries rearrangement is an organic reaction used to convert a phenyl ester to a hydroxy aryl ketone using a Lewis acid catalyst and Brønsted acid work-up. In this reaction, an acyl group belonging to phenol ester migrates to the aryl ring either in the ortho or para position, depending on the reaction temperature and solvent. A low reaction temperature favors para substitution, and with high temperatures, the ortho product dominates [1-2].
The history of this reaction goes back to 1908 when German chemist Karl Theophil Fries reported in a journal.
Examples of Fries Rearrangement
This reaction is used to convert phenyl acetate to ortho- and para- hydroxyphenyl acetate [2].
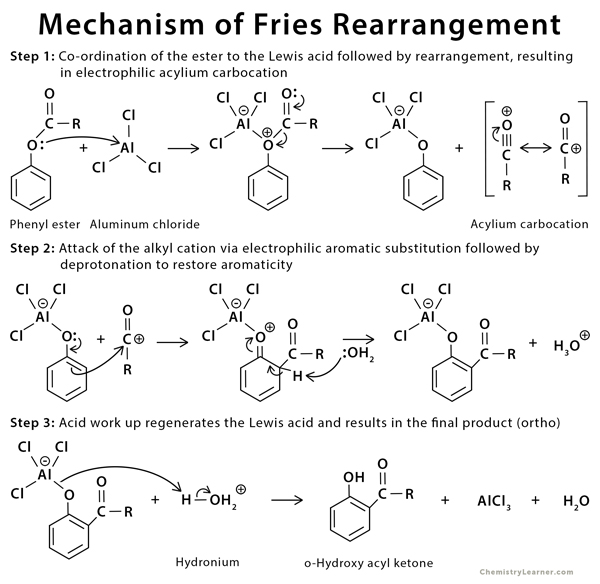
Mechanism of Fries Rearrangement
The reaction proceeds through the formation of a carbocation intermediate [2-4].
Applications of Fries Rearrangement
Fries rearrangement has the following applications:
- Synthesis of o- and p-hydroxy acetophenone, which are useful intermediates in the manufacture of pharmaceuticals
- Synthesis of drug and agrochemical intermediates, thermographic materials, and active antiviral agents
References
- Definition and mechanism – Name-reaction.com
- Definition, example, and mechanism – Chem.libretexts.org
- Mechanism – Researchgate.net
- Mechanism – Organic-chemistry.org