Friedel-Crafts Acylation
What is a Friedel-Crafts Acylation
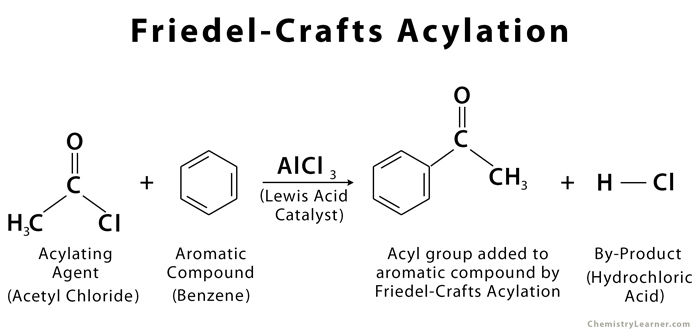
The Friedel-Crafts acylation, also called Friedel-Crafts alkanoylation, is a process that involves the addition of an acyl group to an aromatic compound (benzene), with acyl halide (RCOCl) and aluminum trichloride (AlCl3) being the typical acylating agent and Lewis acid catalyst respectively [1]. Acyl anhydrides can also be used as an acylating agent while iron (iii) chloride can act as a catalyst [2]. It is named after the French chemist Charles Friedel and American chemist James Crafts who developed the reaction in 1877 [1, 2].
Friedel-Crafts Acylation Mechanism with Example
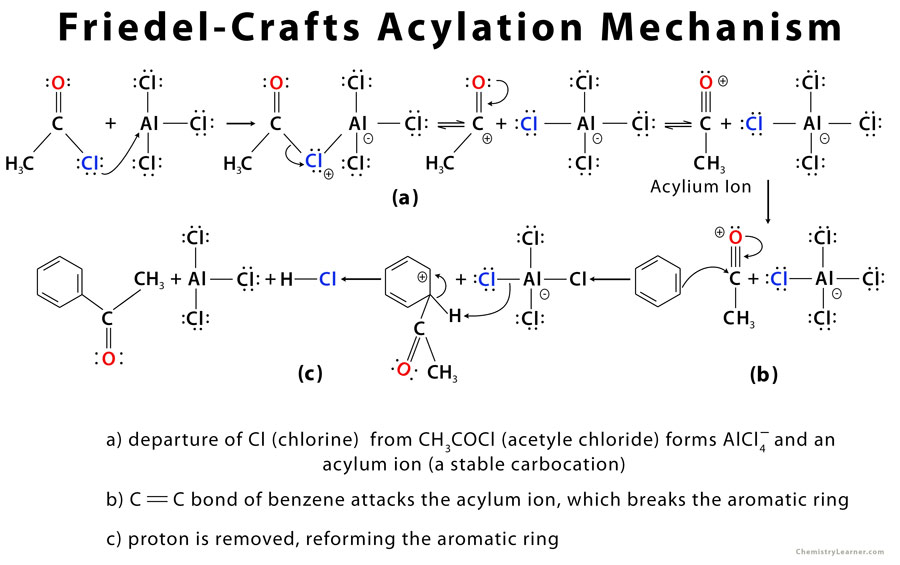
The driving force behind the FC acylation is the activation of electrophile using a Lewis acid to form an acylium ion which is attacked by the aromatic ring [2].
Since the acylium ion is resonance stabilized (as shown in the first step), no rearrangement of carbocation occurs [3]. Moreover, the aromatic compound is deactivated and can no longer accept electrons from a nucleophile (electrophilic attack), preventing it from taking part in further reactions [3].
Practical Applications of Friedel-Crafts Acylation
- Synthesis of natural products and complex bioactive molecules, including africanol, phomazarin, salvadione, bruguierol C, and murrayazoline [4]
- Biological compounds in humans like DNA and vitamin D utilize Friedel-Crafts acylation to attach to their aromatic portions [5]
- Production of phenol and naphthalene using titanium(IV) chloride-mediated ortho-acylation process [6]
- Synthesis of several organic compounds like ferrocene, chlorobenzene, and benzaldehyde [7, 8, 9]
Limitations of Friedel-Crafts Acylation
- It can only be used to produce ketones in which an oxygen atom is joined to a carbon atom by a double bond [1].
- Deactivated benzenes do not react under Friedel-Crafts conditions, as they are always less reactive than the original mono-halobenzene [1].
- The Lewis acid AlCl3 usually forms aryl amine complexes and makes them unreactive [1].
- References
- Friedel-Crafts Acylation of Benzene – Chem.ucalgary.ca
- EAS Reactions (3) – Friedel-Crafts Acylation and Friedel-Crafts Alkylation – Masterorganicchemistry.com
- Friedel-Crafts Acylation – Chem.libretexts.org
- Applications of Friedel-Crafts Reactions in Total Synthesis of Natural Products – Pubs.rsc.org
- Friedel-Crafts Acylation Reactions in Synthesis – Study.com
- Friedel-Crafts Acylation – Organic-chemistry.org
- Friedel-Crafts Acylation of Ferrocene – Cactus.dixie.edu
- Catalytic Friedel-Crafts Acylation of Benzene, Chlorobenzene, and Fluorobenzene using a Novel Catalyst System, Hafnium Triflate and Trifluoromethanesulfonic Acid – Sciencedirect.com
- Friedel-Crafts-Type Reaction of Benzaldehyde with Benzene, Dipronated Benzaldehyde as the Reactive Intermediate – Pubs.acs.org