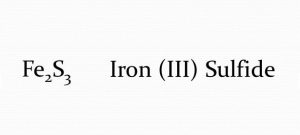Iron(III) Sulfide
Iron(iii) sulfide also called ferric sulfide or iron sesquisulfide is one of the members of iron(3+) sulfides with IUPAC name iron(3+) trisulfide and chemical formula Fe2S3 [1, 2]. It is a relatively unstable artificial black solid that decomposes and turns into a yellow-green powder at room temperature [2].
Iron(III) Sulfide Identification |
|
| CAS Number | 12063-27-3 [1] |
| PubChem CID | 160957 [1] |
| ChemSpider ID | 141408 [3] |
| EC Number | 234-367-5 [1] |
| ChEBI ID | CHEBI:75899 [2] |
| InChI Key | KAEAMHPPLLJBKF-UHFFFAOYSA-N [1] |
Composition and Synthesis
It is produced by the addition of cooled iron(iii) chloride solution to refrigerated sodium sulfide solution, as represented by the following equation:
2FeCl3 + 3Na2S → Fe2S3 + 6NaCl
It can also be synthesized as a by-product of desulfurization of hydrogen sulfide gas that is passed through hydrated iron(iii) oxide:
Fe2O3 + 3H2S + H2O → Fe2S3 + 4H2O
Reaction with Hydrogen Chloride
It undergoes a decomposition reaction when combined with hydrochloric acid [4]:
Fe2S3 + 4HCl → 2FeCl2 + 2H2S + S
Properties and Characteristics of Iron(III) Sulfide
General Properties |
|
| Molar Mass/Molecular Weight | 207.87 g/mol [1] |
Physical Properties |
|
| Color and Appearance | Turns into yellowish-green powder at room temperature [5] |
| Melting Point | Decomposes |
| Boiling Point | N/A [5] |
| Density | 4.3 g cm-3 [5] |
| State of matter at room temperature | Solid [1] |
| Solubility | Moderately soluble in acids |
| Solubility in Water | Very slightly soluble in water [1] |
Use
As a source of sulfur and iron(ii) sulfide during the decay of ferric sulfide at room temperature (about 20 °C).
- References
- Iron Sesquisulfide – Homework.study.com
- CHEBI:75899 – Iron(3+) Sulfide – Ebi.ac.uk
- Iron(3+) Sulfide (2:3) – Chemspider.com
- Chemical Equation Balancer – Chemicalaid.com
- Ferric Sulfide – Americanelements.com