Metalloids
Definition: What Are Metalloids
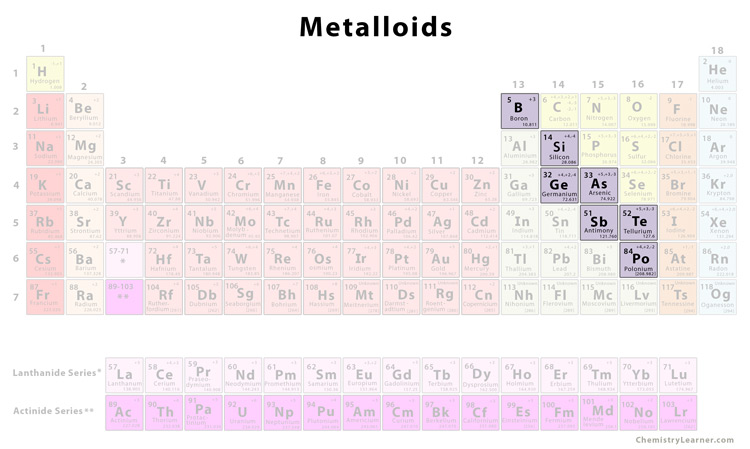
A metalloid is an element having characteristics in between that of metals and non-metals. There are 6 elements in this group [1].
Where are the Metalloids Located on the Periodic Table
The metalloids are found to the right of the post-transition elements and to the left of the non-metals [2].
List of Metalloids |
|
| Boron | B |
| Silicon | Si |
| Germanium | Ge |
| Arsenic | As |
| Antimony | Sb |
| Tellurium | Te |
Polonium (Po) is also commonly considered a metalloid [1]. Other elements included in this list less often include carbon (C), aluminum (Al), selenium (Se) and astatine (At) [3].
Common Properties and Characteristics of Metalloids
Physical Properties
- Shiny like metals, but at the same time brittle like non-metals [4]
- Exist as solids at room temperature
- Fair conductivity at room temperature [3]
Chemical Properties
- Can form alloys with metals [2]
- Metallic or non-metallic behavior in chemical reactions depends upon the substance with which the metalloids are reacting [5]
Generally, metalloids have physical and chemical properties between that metals and non-metals [3].
Difference between Metalloids, Metals, and Non-metals
By definition, metalloids show similar characteristics to both metals and non-metals. Metalloids have a shiny appearance like metals but are brittle like non-metals. In addition to that, their thermal and electrical conductivities fall between metals and non-metals [6].
Chemically, their oxides show amphoteric behavior. Also, their electronegativity and ionization energies are intermediate to those of metals and non-metals [5].
Metalloids as Semiconductors
Due to their intermediate electronic structures, metalloids have enough empty orbitals into which electrons can move, allowing for the conduction of electric current. However, this phenomenon happens only when the metalloid is heated or when small amounts of certain elements are introduced in their crystal lattices (called doping) [1]. These specially treated metalloids are called semiconductors.
Uses of Metalloids
Most of the metalloids find their use in industry. Boron is used as an alloying agent and in pesticides and insecticides [3]. Silicon and Germanium have industrial applications in electronics as semiconductors, while the former is also used in ceramics and glass making [1, 3]. Arsenic finds its use in insecticides, while antimony and tellurium are mainly used as alloying agents [2, 3].
Interesting Facts
- Instead of the standard vertical layout of other families, such as alkali metals and halogens, metalloids are arranged in a diagonal fashion called the ‘stair-step’ or ‘staircase’ [2, 5].
- Silicon is the second most abundant element found in the Earth’s crust [4].
References