Tin
What is Tin
Tin (pronunciation: TIN) is a soft, malleable, silvery-white element classified as a post-transition metal in the periodic table and it is represented by the chemical symbol Sn [1, 2, 3]. It is often mixed with other metals to form alloys, including pewter, solder, Britannia, and many more [4].
Isotopes
Tin has ten naturally-occurring, stable isotopes, the greatest among all the elements, out of which 120Sn is the most abundant with a natural abundance of 32.58% [5]. Among the 29 unstable radioisotopes, only 126Sn has a half-life period of 2.3 X 105 years while the others are short-lived with half-life periods less than one year [5].
Where is Tin Found
It mainly occurs in the reddish, brownish, or yellowish mineral ore cassiterite [1]. Tin is commercially produced through reduction smelting of cassiterite ore by coal in a reverberatory furnace [1, 4]. The top 3 tin-producing countries in the world are China, Indonesia, and Peru while the top 3 reserve-holding nations include China, Indonesia, and Brazil [1].
History
Origin of its Name: The name is derived from an Anglo-Saxon word ‘tin’ whereas its atomic symbol Sn comes from the Latin word ‘stannum’, meaning ‘tin’ [1, 2].
Who discovered it: Although the discovery of tin is unclear, it is believed to have been found by the ancient Egyptians, Sumerians, Chinese, Incas, and the Indus Valley people [1].
When and How was it Discovered
Historical evidence suggests that tin played an important role in human lives mainly because of bronze, an alloy containing roughly 90-95 percent copper and 5-10 percent tin, and was ideal for weapons and tools [1, 3]. Tin was also valued for its properties, as a pilgrim bottle and tin ring were unearthed from an Egyptian tomb that dates to the eighteenth dynasty (approximately from 1580-1350 BC) [1].
It was mined by the ancient Chinese in the Yunnan province roughly during 700 BC [1]. Pure tin has been excavated at Machu Picchu, the fortress of the Incas [1]. The ancient Greeks imported tin by sea and called the source ‘The Cassiterides’, which means Tin Islands [3]. These were probably Cornwall in Great Britain and north-west Iberia in Spain where large tin deposits exist [3].
Tin Identification |
|||
| Atomic number | 50 [1] | ||
| CAS number | 7440-31-5 [1] | ||
| Position in the periodic table | Group | Period | Block |
| 14 [1] | 5 [1] | p [1] | |
Properties and Characteristics of Tin
General Properties |
||||||||||||||||
| Relative atomic mass | 118.710 [1] | |||||||||||||||
| Atomic mass | 118.710 amu [1] | |||||||||||||||
| Molar mass | 118.7100 g/mol [6] | |||||||||||||||
| Allotropes | Gray (alpha) Sn, White (beta) Sn [1] | |||||||||||||||
Physical Properties |
||||||||||||||||
| Color | Silvery-white [3] | |||||||||||||||
| Melting point/freezing point | 231.928 °C, 449.47 °F [1] | |||||||||||||||
| Boiling point | 2586 °C, 4687 °F [1] | |||||||||||||||
| Density | 7.287 g cm-3 [1] | |||||||||||||||
| State of matter at room temperature (solid/liquid/gas) | Solid [1, 3] | |||||||||||||||
| Hardness | ||||||||||||||||
| – Brinell | 51 MPa [7] | |||||||||||||||
| – Mohs | 1.5 [7] | |||||||||||||||
| – Vickers | Unknown [7] | |||||||||||||||
| Electrical Conductivity | 9.1 X 106 S/m [7] | |||||||||||||||
| Charge | +2, +4 [8] | |||||||||||||||
| Thermal (heat) conductivity | 67 W/(m K) [7] | |||||||||||||||
| Specific heat | 227 J kg-1 K-1 [1] | |||||||||||||||
| Bulk modulus | 58.2 GPa [1] | |||||||||||||||
| Shear modulus | 18.4 GPa [1] | |||||||||||||||
| Young’s modulus | 49.9 GPa [1] | |||||||||||||||
| Vapor pressure | ||||||||||||||||
| – Temperature (K) | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 | 2200 | 2400 | |||||
| – Pressure (Pa) | – | – | 1.26 X 10-9 | 1.26 X 10-6 | 3.1 X 10-3 | 0.207 | 4.85 | 56.3 | – | – | – | |||||
Chemical Properties |
||||||||||||||||
| Oxidation state/Oxidation number | −4, +2, +4 [1] | |||||||||||||||
| Isotopes | Isotope | Mass | Abundance (%) | Half-life | Mode of decay | |||||||||||
| 112Sn | 111.905 | 0.97 | – | – | ||||||||||||
| 114Sn | 113.903 | 0.66 | – | – | ||||||||||||
| 115Sn | 114.903 | 0.34 | – | – | ||||||||||||
| 116Sn | 115.902 | 14.54 | – | – | ||||||||||||
| 117Sn | 116.903 | 2.8 | – | – | ||||||||||||
| 118Sn | 117.902 | 24.22 | – | – | ||||||||||||
| 119Sn | 118.903 | 8.59 | – | – | ||||||||||||
| 120Sn | 119.902 | 32.58 | – | – | ||||||||||||
| 122Sn | 121.903 | 4.63 | – | – | ||||||||||||
| 124Sn | 123.905 | 5.79 | > 2.2 X 1018 y | β-β- | ||||||||||||
Atomic Data of Tin (Element 50)
| Valence electrons | 4 [9] | |||||||
| Quantum numbers | ||||||||
| – n | 5 [9] | |||||||
| – ℓ | 1 [9] | |||||||
| – mℓ | 0 [9] | |||||||
| – ms | +1/2 [9] | |||||||
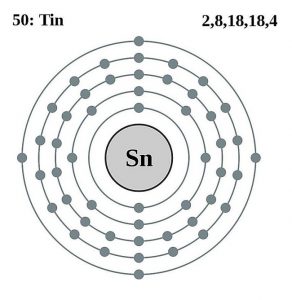
| Electron configuration (noble gas configuration) | [Kr] 4d105s25p2 [1] | |||||||
| Atomic structure | ||||||||
| – Number of electrons | 50 [3] | |||||||
| – Number of neutrons | 68 [3] | |||||||
| – Number of protons | 50 [3] | |||||||
| Radius of Atom | ||||||||
| – Atomic radius | 2.17 Å [1] | |||||||
| – Covalent radius | 1.40 Å [1] | |||||||
| Electronegativity (Pauling-scale) | 1.96 [1] | |||||||
| Electron affinity | 107.298 [1] | |||||||
| Ionization energy (kJ mol-1) | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
| 708.581 | 1411.793 | 2943.054 | 3930.332 | 6973.96 | – | – | – | |
Tin Electron Configuration (Bohr Model)
What is it Used for
- Tin is used as a high-polishing compound to coat other metallic surfaces for preventing corrosion, like in tin cans (tin drums), tin roofs, and tin ceiling tiles, which are made out of tin-coated steel [1, 3]. Spraying tin salts onto glass produces electrically-conductive coatings [1].
- Commercially important tin alloys such as niobium-tin is used for producing superconducting magnets [1].
- Molten glass is poured onto molten tin for manufacturing flat surfaces of window glass [1].
- It can be rolled into thin sheets called ‘tinfoil’, which is used for covering or wrapping food [3]. Today, it is typically produced from aluminum [3].
- Tin (II) chloride is a reducing agent that is used for dyeing silk and calico while tin (IV) oxide is used in gas sensors and ceramics [1]. Another zinc compound, zinc stannate (Zn2SnO4), is used in plastics because of its fire-retarding properties [1].
- Tin compounds are commercially used in anti-fouling paint that is applied to the hull of ships and boats for preventing barnacles [1]. However, the compound has been found to be toxic to marine life and has now been replaced by innovative paints that are free from tin and copper [1].
- Tin fluoride (SnF2) is an essential ingredient in some toothpaste [3].
Tin Toxicity
Since they enter and leave the body rapidly after inhaling or eating them, the metallic or inorganic tin compounds do not cause harm to the human body and are considered non-toxic [1, 10]. However, inhalation, eating, drinking, or skin contact with organotin compounds can cause skin, eye, and respiratory irritation, neurological problems, and gastrointestinal poisoning [10]. It does not play any biological role in the human body [1].
Interesting Facts
- The element is graphically represented by an image of a tin-coated steel can, marked with an alchemical sign for tin [1].
- Bending tin at room temperatures causes a high-pitched creaking noise, commonly called the “tin cry”, due to the change in shape of tin’s crystal structure [4].
- It turns into the powdery alpha-tin allotropic form below 13° C [4].
- The Britannia metal, an alloy of tin, is actually used in making the golden statuettes that are presented to the various category winners at the Academy Awards [4].
- The beta allotropic form of tin is paramagnetic, which means it does not have its own magnetic field but can be magnetized by external magnetic fields [11].
Cost of Tin
The price of pure tin is about $0.24 per gram, and in bulk, it costs about $0.018 per gram [3].
- References
- http://www.rsc.org/periodic-table/element/50/tin
- https://education.jlab.org/itselemental/ele050.html
- https://www.chemicool.com/elements/tin.html
- https://www.livescience.com/37355-tin.html
- https://education.jlab.org/itselemental/iso050.html
- https://www.webqc.org/molecular-weight-of-Sn.html
- http://periodictable.com/Elements/050/data.html
- Gpb.org
- http://chemistry-reference.com/q_elements.asp?Symbol=Sn
- https://www.atsdr.cdc.gov/phs/phs.asp?id=541&tid=98
- https://sciencing.com/tin-cans-attracted-magnet-7422918.html