Curium
What is Curium
Curium (pronounced as KYOOR-ee-em) is a radioactive metal belonging to the family of actinide metals and denoted by the chemical symbol Cm [1].
Curium Isotopes
It has 15 isotopes out of which the most stable one is Curium-247 with a half-life of about 15,600,000 years that undergoes alpha-decay to form plutonium-243 [3].
History
Origin of its Name: It is named after the well-renowned scientists, Pierre Curie and Marie Curie, the discoverers of the radium.
Who Discovered it: The element was synthesized by the research team of Glenn Seaborg, Albert Ghiorso, and Ralph James [1].
Where was Curium Discovered
In 1944, Seaborg, Ghiorso, and James bombarded plutonium-239 with alpha-particles in a cyclotron at Berkeley, California. It was then sent to the Metallurgical Laboratory at the University of Chicago to isolate and analyze the new element. On 11 November 1945, after the end of the World War 2 the discovery was revealed by Seaborg at a radio show where he had been invited as the guest scientist. It was made official the following week [1].
Classification and Position of the Element on the Periodic Table [1]
| Group Number | Unknown |
| Period | 7 |
| Block | f |
Properties of Curium [1, 2, 3, 5]
General Properties |
|||
| Relative atomic mass | 247 | ||
| Atomic mass | 247 atomic mass units [1] | ||
Physical Properties |
|||
| Color/appearance | Silver white | ||
| Luster | Unknown | ||
| Odor | Unknown | ||
| Melting point/freezing point | 1345 °C (2453°F) | ||
| Boiling Point | Unknown | ||
| Density | 13.51 g/cm3 | ||
| State of matter at room temperature (solid/liquid/gas) | Solid | ||
| Hardness | Unknown | ||
| Electrical conductivity | Unknown | ||
| Thermal (heat) conductivity | Unknown | ||
| Specific heat | Unknown | ||
Chemical Properties |
|||
| Flammability | Unknown | ||
| Oxidation state/Oxidation number | +3, +4 | ||
Atomic Data of Curium [1, 2, 3]
| Atomic number | 96 | ||||||
| Valence electrons | 2 | ||||||
| Quantum numbers | |||||||
| – n | 5 | ||||||
| – ℓ | 3 | ||||||
| – m ℓ | -3 | ||||||
| – m s | -1/2 | ||||||
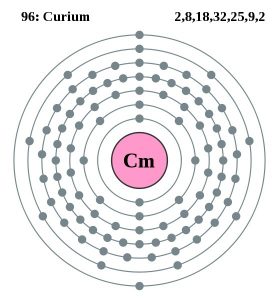
| Electron configuration (noble gas configuration) | [Rn] 5f76d17s2 | ||||||
| Atomic structure | |||||||
| – Number of electrons | 99 | ||||||
| – Number of neutrons | 151 | ||||||
| – Number of protons | 99 | ||||||
| Radius of Atom | |||||||
| – Atomic radius | 2.45 Å | ||||||
| – Covalent radius | 1.68 Å | ||||||
| Electronegativity | Unknown | ||||||
| Ionization energy
(kJmol-1) |
1st | 2nd | 3rd | 4th | 5th | 6th | 7th |
| 578.082 | – | – | – | – | – | – | |
Curium Bohr Model
What is Curium Used for
As it is not found naturally in the earth’s crust, the radioactive element has only been used in basic research studies due to its limited laboratory production. Also, it does not react with other compounds. However, curium-244 might be applicable as a power source for operating radioisotope thermoelectric generators used in spacecraft [2, 3].
Interesting Facts About Curium
- Curium-244 has been used in Alpha Proton X-ray Spectrometer to detect the presence of chemical elements in the atmosphere and rock surface compounds of Mars [3, 4]
- The element is believed to glow red in the dark due to its radioactive nature [3]
How Much Does Curium Cost
The radioactive element is not commercially available outside of laboratory production.
- References