Phenol
Phenol or carbolic acid is a chemical compound that belongs to the class of organic compounds known as phenols. It is fundamental in producing many essential chemicals, such as plastics, resins, dyes, detergents, and pharmaceuticals.
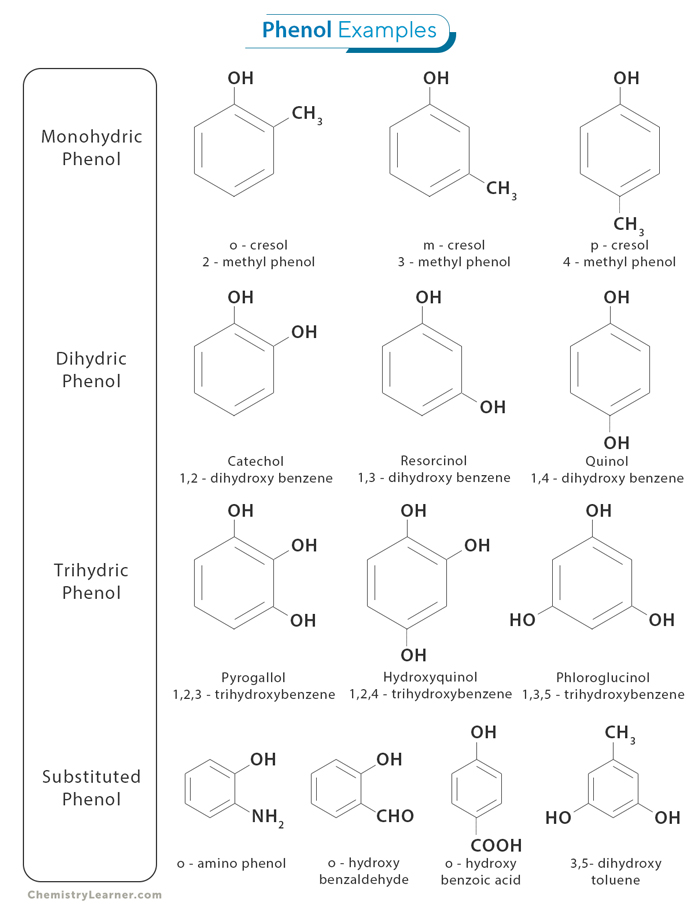
While phenols share some similarities with alcohols due to the presence of the hydroxyl group, their distinct structural and chemical properties make them a separate class of compounds with unique characteristics and behaviors. [1-4]
Formula and Structure
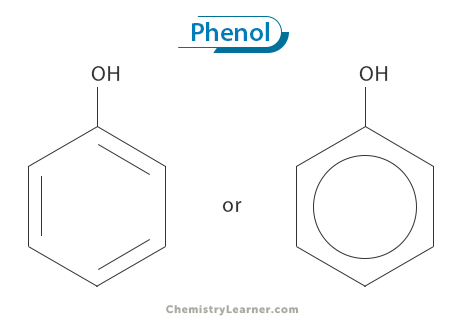
Phenol’s molecular formula is C6H6O. It is also known as hydroxybenzene due to its benzene ring structure, which consists of six carbon atoms connected in a cyclic arrangement. One of these carbon atoms is bonded to a hydroxyl group (-OH), replacing one hydrogen atom on the benzene ring. [1-4]
Physical Properties
Phenol displays unique physical properties that are listed below. [1-4]
Physical State: Phenol is a white crystalline solid at room temperature but can also be a colorless liquid.
Odor: Phenol has a distinctive, sweet, and medicinal odor.
Melting Point: The melting point of phenol is around 40.5 °C (104.9 °F).
Boiling Point: Phenol has a relatively high boiling point of approximately 181.7 °C (359.1 °F).
Density: The density of liquid phenol is around 1.07 g/cm³.
Solubility: Phenol is partially soluble in water. However, it is more soluble in organic solvents like ether, acetone, and chloroform.
Is Phenol an Acid or Base
Phenol acts as a weak acid. The hydroxyl group can release a proton (H+) from the oxygen atom, making phenol slightly acidic in aqueous solutions. The resulting equilibrium reaction in water can be represented as: [1-4]
C6H5OH ⇌ C6H5O– + H+
As a weak acid, phenol forms phenolate ions in solution. It has an acid dissociation constant (pKa) value of around 10, which is higher than many common acids, making it a weaker acid in comparison.
Synthesis
The synthesis of phenol can be achieved through several methods, each involving introducing a hydroxyl (-OH) group onto an aromatic ring. [1-4]
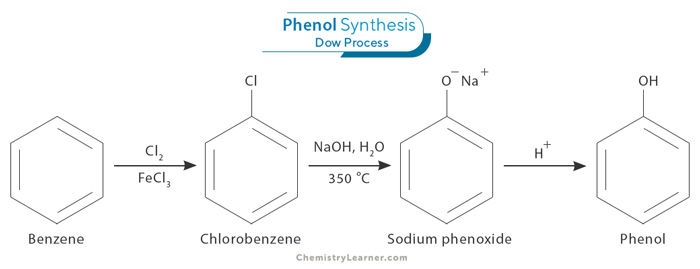
1. Hydrolysis of Chlorobenzene (Dow Process)
Dow process starts with the preparation of chlorobenzene (C6H5Cl) from electrophilic aromatic substitution, where chlorine (Cl2) replaces a hydrogen atom in benzene (C6H6) with the help of an iron (III) chloride (FeCl3) catalyst. It then reacts with a concentrated sodium hydroxide (NaOH) solution at high temperatures and pressures to form sodium phenoxide (C6H5O–Na+), which is then acidified to yield phenol.
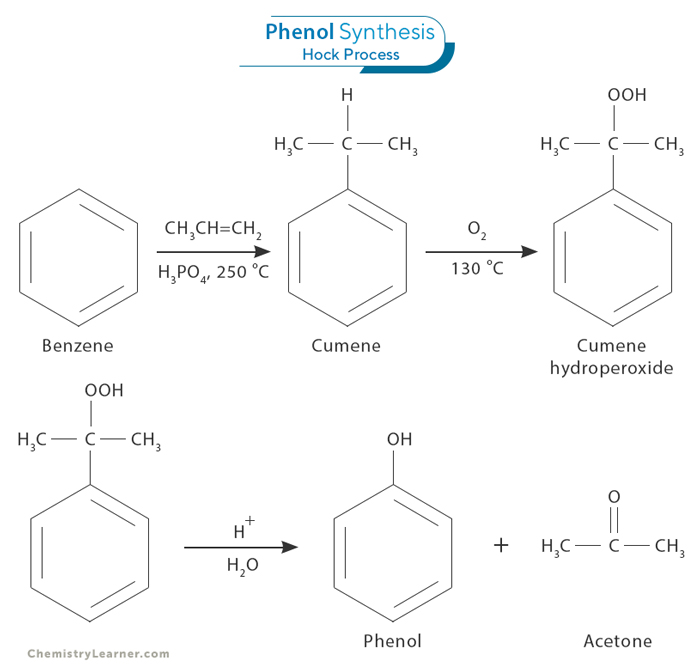
2. Oxidation of Isopropylbenzene (Hock Process)
The Hock process involves the conversion of benzene and propylene to isopropylbenzene (cumene), followed by oxidation to produce phenol. Acetone is formed as a byproduct. This process has gained popularity due to its high efficiency and cost-effectiveness.
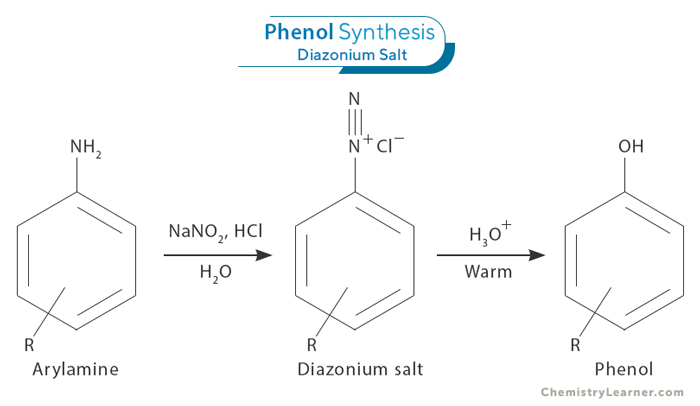
3. Diazotization of Arylamine
A third method of synthesizing phenol involves the diazotization of an arylamine. The arylamine is first treated with sodium nitrite and hydrochloric acid to form the corresponding diazonium salt. This diazotization step is crucial as it allows for forming a diazonium salt from an arylamine precursor. Once the diazonium salt is formed, it can undergo hydrolysis to yield phenol and release nitrogen gas.
Chemical Reactions
The hydroxyl group imparts specific chemical properties to phenol, making it undergo various chemical reactions that play a significant role in organic synthesis and industrial processes. [1-4]
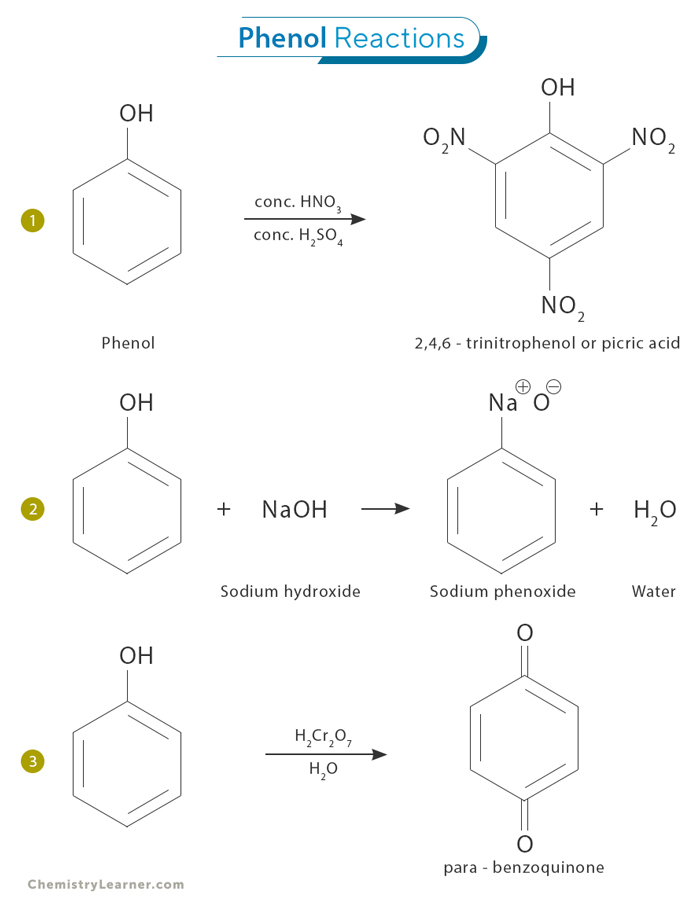
1. Electrophilic Aromatic Substitution
Electrophilic aromatic substitution reactions take on a distinctive character on phenol’s unique aromatic character consisting of a hydroxyl (-OH) group directly attached to a benzene ring. For example, nitration involves the introduction of a nitro group (-NO2) onto the phenol ring, often using a mixture of concentrated nitric acid (HNO3) and concentrated sulfuric acid (H2SO4) as the nitrating agents. The hydroxyl group in phenol activates the aromatic ring toward electrophilic attack by enhancing its electron density. Consequently, the nitronium ion (NO2+) readily attacks the activated phenol ring, forming 2,4,6 – trinitrophenol.
2. Reaction with Base
When phenol reacts with a base, such as sodium hydroxide (NaOH), it undergoes deprotonation. The hydroxyl group in the phenol molecule donates its proton to the base, forming water and the corresponding phenoxide ion. This reaction is commonly known as neutralization. Phenoxide ion is highly reactive and reacts with NaOH to form sodium phenoxide, a colorless solution.
3. Oxidation Reactions
Phenols can be oxidized by oxidizing agents such as potassium permanganate (KMnO4) or chromic acid (H2CrO4). Oxidation typically involves the loss of electrons from the phenol molecule, leading to various products depending on reaction conditions. One of the products formed is the dicarbonyl compound para-benzoquinone, also known as 1,4-benzoquinone.
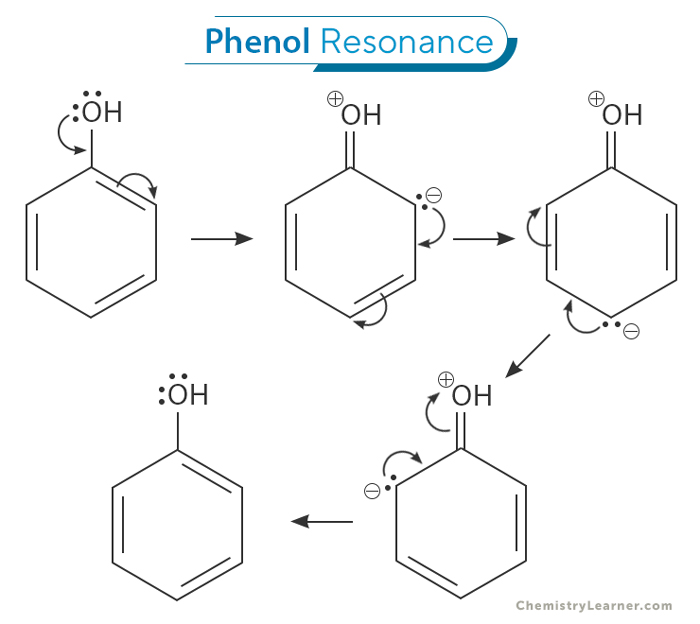
Phenolate Resonance Structures
Phenol exhibits fascinating resonance structures due to its unique electron distribution. The oxygen atom in the hydroxyl group is more electronegative than the carbon atom it is bonded to, resulting in a partial negative charge on the oxygen and a partial positive charge on the carbon. This charge separation gives rise to a resonance structure in which the pi electrons of the benzene ring are delocalized, with the negative charge distributed across the adjacent carbon atom. The resonance contributes to the overall stability of phenol and its enhanced reactivity in various chemical reactions. [1-4]
Uses and Applications
Phenols have found diverse applications in various industries due to their unique properties. [1-4]
Antiseptic and disinfectant: Phenols are used as antiseptics and disinfectants to kill or inhibit the growth of microorganisms. They are often found in mouthwashes, throat sprays, and disinfectant solutions.
Preservatives: Phenolic compounds are used as preservatives in various industries, such as food preservation and cosmetics, to extend the shelf life of products by preventing the growth of bacteria and fungi.
Pharmaceuticals: Phenols are used to synthesize pharmaceutical drugs, including analgesics, antiseptics, and antipyretics. They can also be used as intermediates in producing other pharmaceutical compounds. Phenol derivatives like salicylic acid are valuable in over-the-counter medications for treating acne.
Manufacturing resins and plastics: Phenolic resins, such as Bakelite, are used in producing various molded products, including electrical insulators and kitchen utensils, due to their heat-resistant and insulating properties. Phenolic compounds are also used to make thermosetting plastics for consumer goods.